Endo F2
Endo F2 is a highly specific recombinant endoglycosidase which cleaves within the chitobiose core of asparagine-linked complex biantennary and high mannose oligosaccharides from glycoproteins and glycopeptides. Endo F2 cleaves biantennary glycans at a rate approximately 20 times greater than high mannose glycans. The activity of Endo F2 is identical on biantennary structures with and without core fucosylation. However, Endo F2 is not active on hybrid or tri- and tetra-antennary oligosaccharides.
- Endo F2 is tagged with a chitin binding domain (CBD) for easy removal from a reaction
- Recombinant enzyme with no detectable exooglycosidase or other endoglycosidase contaminating activities
- Glycerol -free for optimal performance in HPLC and mass spectrometry analysis
- ≥95% purity, as determined by SDS-PAGE and intact ESI-MS
- Optimal activity and stability for up to 24 months

-
Product Information
Specificity:
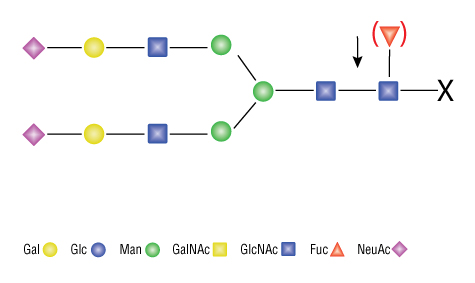
Product Source
Cloned from Elizabethkingia miricola (formerly Flavobacterium meningosepticum) and expressed in E. coli.- This product is related to the following categories:
- Endoglycosidases
- This product can be used in the following applications:
- Glycan Sequencing
-
Protocols, Manuals & Usage
-
Tools & Resources
-
FAQs & Troubleshooting
-
Citations & Technical Literature
-
Quality, Safety & Legal
Ineligible item added to cart
Based on your Freezer Program type, you are trying to add a product to your cart that is either not allowed or not allowed with the existing contents of your cart. Please review and update your order accordingly. If you have any questions, please contact Customer Support at freezers@neb.com or 1-800-632-5227 x 8.









