Bacteroides Heparinase I
Bacteroides Heparinase I, also called Heparin Lyase I, is active on heparin and the highly sulfated domains of heparan sulfate. The reaction yields oligosaccharide products containing unsaturated uronic acids which can be detected by UV spectroscopy at 232 nm.
- Recombinant enzyme with no detectable glycosidase, sulfatase or uronidase contaminating activities
- ≥95% purity, as determined by SDS-PAGE and intact ESI-MS
- Optimal activity and stability for up to 12 months when stored in solution at -80°C
- Optimal for use in coagulation studies

-
Product Information
Bacteroides Heparinase I cloned from Bacteroides eggerthii, also called Heparin Lyase I, is active on heparin and the highly sulfated domains of heparan sulfate. The reaction yields oligosaccharide products containing unsaturated uronic acids which can be detected by UV spectroscopy at 232 nm.
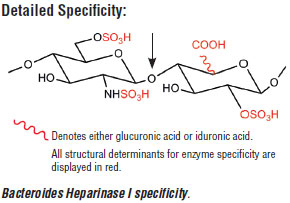
Bacteroides Heparinase I cleaves the glycosidic bond between N-sulfated hexosamines and 2-O-sulfated iduronic acid residues, between these same as well as the 2-O-sulfated glucuronic acid residues. The 2-O-sulfated uronic acid residue is essential for the activity of Bacteroides Heparinase I and 6-O-sulfation of GlcNS does not hinder enzyme activity.
Product Source
Cloned from Bacteroides Eggerthii and expressed in E. coli.- This product is related to the following categories:
- Heparinases Products
- This product can be used in the following applications:
- Analysis of Heparin/HS
-
FAQs & Troubleshooting
-
Citations & Technical Literature
-
Quality, Safety & Legal
Other Products You May Be Interested In
Ineligible item added to cart
Based on your Freezer Program type, you are trying to add a product to your cart that is either not allowed or not allowed with the existing contents of your cart. Please review and update your order accordingly If you have any questions, please contact Customer Service at freezers@neb.com or 1-800-632-5227 x 8.