Monarch® Kits for Plasmid Purification
< Return to NEBMonarch.com
Monarch kits for plasmid purification are currently available in miniprep format for the purification of up to 20 µg of plasmid DNA from bacterial cultures. Buffers and columns are available separately for added convenience and kits are designed with sustainability in mind
Reasons to Choose Monarch Kits for Plasmid Purification
- Unique column design eliminates buffer/salt carryover, allows for elution in ≥ 30 µl, and enables easy handling
- Isolate high-quality, concentrated plasmid DNA of various sizes with excellent yield and purity
- Optimized colored buffer system for monitoring protocol steps (and no need to add additional RNase!)
| SPECIFICATIONS | |
|---|---|
| Culture Volume | 1-5 ml, not to exceed 15 OD units |
| Binding Capacity | up to 20 μg |
| Plasmid Size | up to 25 kb |
| Typical Recovery | up to 20 μg. Yield depends on plasmid copy number, host strain, culture volume, and growth conditions. |
| Elution Volume | ≥ 30 μl |
| Purity | A260/280and A260/230≥ 1.8 |
| Protocol Time | 10½ minutes of spin and incubation time |
| Compatible Downstream Applications |
restriction digestion and other enzymatic manipulations, transformation, transfection, DNA sequencing, PCR, labeling, cell-free protein synthesis, etc. |
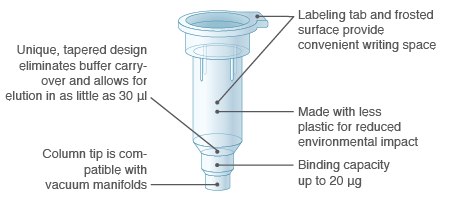
Unique Column Design
The design of Monarch miniprep columns eliminates buffer retention and salt carryover, resulting in highly pure plasmid DNA for your downstream needs. Elution can be done in as little as 30 µl, and convenient tab and frosted surfaces facilitate handling and labeling.
Isolate high-quality, concentrated plasmid DNA
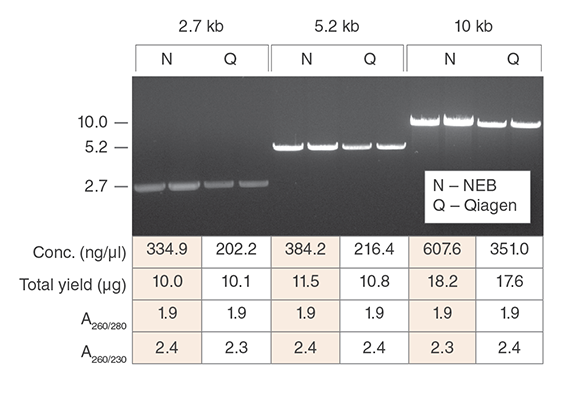
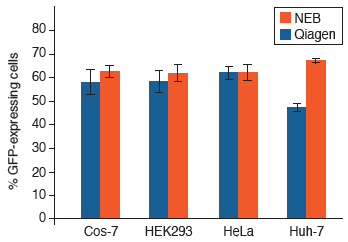
Purify up to 20 µg of plasmid DNA of various sizes with consistently high purity and quality, ready for use in downstream application including digestion, cloning, PCR and transfection of robust cell lines.
Optimized Buffer System
Colored buffers prevent confusion and allow for monitoring of certain steps in the protocol. RNase is provided as part of the neutralization buffer, so there is no need to add it before starting, and no risk of forgetting to add it.
Less buffer retention as advertised! The indicator system prevents under and over-mixing for lysis and neutralization step, minimizing genomic DNA fragmentation.




