Strand Displacement Amplification & Nicking Enzyme Amplification Reaction
Strand displacement amplification (SDA) relies on a strand displacement DNA polymerase and a DNA nicking event targeted via primer design and a nicking endonuclease. The nicking site is regenerated with each polymerase displacement step for repeated cycles of nicking and extension, with the downstream strand displaced and free to anneal to primers in solution for amplification from the other end, resulting in exponential amplification. The nature of the SDA reaction produces discrete fragments of DNA that can easily be utilized in downstream workflows, though some fragments will vary by the presence/absence of the nicking site in the product.
| Reaction Temperature | Amplicon Size | Detection Method(s) |
|---|---|---|
| 60°C | <100 nt | Fluorescence |
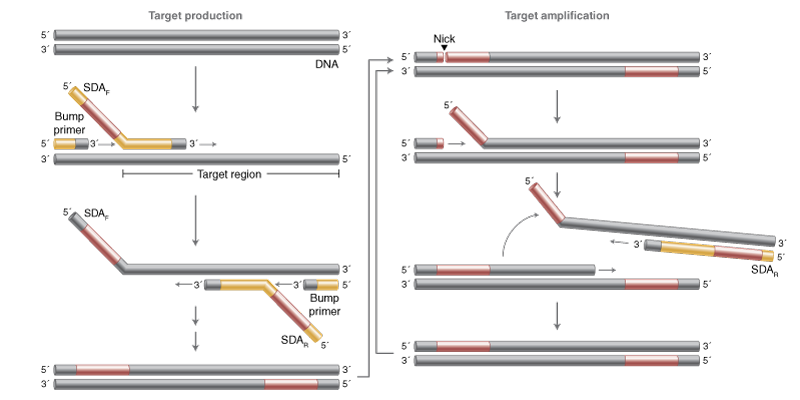
SDA utilizes two outer “bump” primers and two inner primers with 5’ tail regions that contain a nicking enzyme recognition site. In conjunction with a nicking enzyme (e.g., NtBstNBI), amplification of discrete DNA products occurs in rapid fashion.
DNA Strand Displacement
Early versions of SDA used Klenow (exo-) DNA polymerase and a nucleotide analog such as an α-thiol dCTP to prevent restriction enzyme cleavage of the amplified strand, thus generating a single stranded nick. The development of nicking enzymes such as Nt.BstNBI and WarmStart Nt.BstNBI have enabled simpler versions of this method. When combined with a Bst polymerase, extremely rapid amplification can be achieved.
Like other amplification techniques, SDA can be affected by carryover contamination, where amplified DNA from a previous reaction can serve as the unintended template for future rounds. One way to mitigate this is by the inclusion of dUTP and WarmStart Afu Uracil-DNA Glycosylase (UDG). WarmStart Afu UDG contains an aptamer that inhibits enzyme activity at temperatures below 42°C, enabling room temperature setup and low temperature amplification such as SDA. Following amplification, the temperature is increased (>50°C) causing dissociation of the aptamer from the enzyme and the resultant destruction of dU-containing amplicons, thus preventing carryover into the next reaction.
When designing SDA reactions, 4 primers are needed: two outer “bump” primers to provide the initial displacement of target sequence (generally a short <100 bp region), and two inner primers that include 5′ tail regions containing a nicking enzyme recognition site (e.g., Nt.BstNBI). It is generally useful to separate the nicking site from the target sequence by a 4-dA spacer as outlined in Ehses et al., which includes example primer tails.
Most PCR primer design software can be used to aid in SDA primer design. The NEB LAMP Primer Design Tool (typically for LAMP primer design) can also be used with modifications.* Reactions can be detected with real-time fluorescence using an intercalating dye or probe.
Nicking Enzyme Amplification Reaction
A similar amplification approach, the Nicking Enzyme Amplification Reaction (NEAR), powers point-of-care detection platforms that offer sample-to-answer results in minutes. This technique was originally described by Van Ness, J et al. and recently reviewed by Qain, C et al.
*When using LAMP primer design software, use the F3/B3 regions as Bump primers. For the tailed primers, take only the F2/B2 sequences from the inner primers, and add SDA tails where the F1c/F2c would be.
Choose Type:
- Can Bst 2.0® DNA Polymerase initiate at a nick in the DNA?
- Can Bst 2.0® DNA Polymerase be used in labeling reactions and partial fill in reactions?
- What is the activity of nicking enzymes at different temperatures?
- What advantages does WarmStart® Nt.BstNBI provide?
- How can WarmStart Afu Uracil-DNA Glycosylase (UDG) be used for carryover prevention?
- What does WarmStart mean?
- Isothermal Amplification Brochure
- Amplification Reagents for Molecular Diagnostics Applications (2017)
Brochures
Posters
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. All other trademarks are the property of their respective owners. The use of this product may require the buyer to obtain additional third-party intellectual property rights for certain applications. For more information, please email busdev@neb.com.
This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.



