Protein Localization
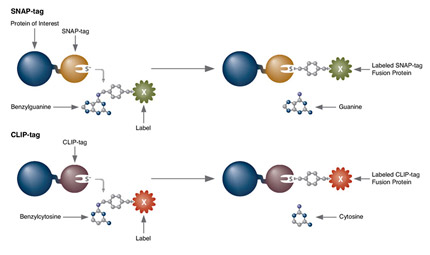
Protein functional activities correspond with their subcellular expression and molecular complexing interactions. Localization can be effectively demonstrated with fluorescence microscopy based techniques or fractionation procedures. A broad spectrum of fluorescence imaging can be accomplished by using recombinant reporter proteins, (i.e. GFP, SNAP-tag®), or fluorescent dyes (i.e. Alexa), or fluorophore labeled molecules (i.e. protein specific antibodies). Cells can be genetically modified to overexpress protein targets, or regulatory components of non-coding sequences, for purposes such as determining fundamental cellular processes, disease mechanisms, gene therapy, and response to therapies. Fusion protein tags can be detected by antibodies or have functional properties to enable localization. Bioluminescent protein and fluorescent protein labeling systems are such genetically engineered optical imaging tools, with greater specificity at lower concentrations than other methods. With this approach, imaging can be in fixed or living cells, tissues or animals. A very attractive feature of the labeling of fusion proteins is that the labeling itself can be restricted to certain locations of a cell (i.e. SNAP-Cell® or SNAP-Surface®). Such discrimination cannot be easily achieved when using bioluminescent proteins. Some fluorescent protein labeling systems enable small nonfluorescent molecules to become fluorescent when bound to a small genetically inserted peptide sequence in the target protein of interest (i.e. FlAsH). Advances in genetically engineered fluorescence systems and microscopy optic machinery, have made imaging a core method for protein localization.
SNAP-tag®, SNAP-Cell® and SNAP-Surface® are registered trademarks of New England Biolabs, Inc.
Choose Type:
- Cellular Labeling (E9100)
- Cellular Labeling (E9230)
- Cellular Labeling (S9103)
- Protocol for Cellular Labeling with SNAP-Cell® Oregon Green® (NEB #S9104)
- SNAP-Cell® TMR-Star Cellular Labeling (NEB #S9105)
- Cellular Labeling (S9107)
- Cellular Labeling (S9109)
- Cellular Labeling (S9110)
- Cellular Labeling (S9112)
- Cellular Labeling (S9124)
- Cellular Labeling (S9129)
- Cellular Labeling (S9132)
- Cellular Labeling (S9134)
- Cellular Labeling with SNAP-Surface® Alexa Fluor® 647 (NEB #S9136)
- Cellular Labeling (S9217)
- Cellular Labeling (S9219)
- Cellular Labeling (S9232)
- Cellular Labeling (S9233)
- Cellular Labeling (S9234)
- Cloning of CLIP-tag Fusions in pCLIPf (N9215)
- CoA 488 (S9348)
- Expression of CLIP-tag Fusions (N9215)
- Instructions for Use with ACP Synthase (P9301)
- Instructions for Use with SFP Synthase (P9302)
- Labeling of Proteins in vitro (S9110)
- Labeling of Proteins in vitro (P9302)
- Labeling of Proteins in vitro (S9103)
- Labeling of Proteins in vitro (S9104)
- Labeling of Proteins in vitro (S9105)
- Labeling of Proteins in vitro (S9106)
- Labeling of Proteins in vitro (S9107)
- Labeling of Proteins in vitro (S9109)
- Labeling of Proteins in vitro (S9143)
- Labeling of Proteins in vitro (S9217)
- Labeling of Proteins in vitro (S9219)
- Labeling of Proteins in vitro (S9220)
- Labeling of Proteins in vitro (S9221)
- Labeling of Proteins in vitro (S9232)
- Labeling of Proteins in vitro (S9233)
- Labeling of Proteins in vitro (S9234)
- Labeling of Proteins in vitro (P9301)
- Instructions for Labeling of Proteins in vitro (S9348)
- Labeling of Proteins in Solution (E9230)
- Labeling of Proteins in vitro (S9351)
- Labeling on the Surface of Cells (S9349)
- Labeling on the Surface of Cells (S9350)
- Labeling on the Surface of Cells (S9351)
- Labeling Proteins in vitro (S9112)
- Labeling Proteins in vitro (S9124)
- Labeling Proteins in vitro (S9129)
- Labeling Proteins in vitro (S9132)
- Labeling Proteins in vitro (S9134)
- Labeling Proteins in vitro (S9136)
- Use of SNAP-Cell Block with SNAP-Cell Substrates (E9100)
- Use of CLIP-Cell Block with CLIP-Cell Substrates (E9230)
- Use with CLIP-tag substrates (S9220)
- View the video "Fluorescent Labeling of COS-7 Expressing SNAP-tag Fusion Proteins for Live Cell Imaging" in the Journal of Visualized Experiments (JoVE)
- Western Analysis (E8023)
- Cellular Labeling (E9120)
- Instructions for Cellular Labeling (E9200)
- Labeling Proteins in vitro (E9120)
- Labeling Proteins in vitro (E9200)
- Cloning of SNAP-tag Fusions in pSNAPf (N9183)
- Cloning of SNAP-tag Fusions in pSNAP-tag(T7)-2 (N9181)
- Expression of SNAP-tag Fusions (N9181)
- Expression of SNAPf Fusions (N9183)
- Labeling of Proteins in vitro (S9349)
- Labeling of Proteins in vitro (S9350)
- Cellular Labeling (S9221)
- Labeling of Proteins in vitro (E9100)
- Use with SNAP-Surface substrates (S9143)
- Use with SNAP-Cell Substrates (S9106)
- Cellular Labeling (S9159)
- Labeling of Proteins in vitro (S9159)
- SNAP-Cell® 647-SiR Cellular Labeling (NEB #S9102)
- Labeling of Proteins in vitro (S9102)
-
SNAP-tag® Technologies: Tools to Study Protein Function
Read about the NEB’s set of protein tools for the specific labeling (SNAP-, CLIP-, ACP- and MCP-tags) of fusion proteins.
- Cellular Imaging & Analysis Brochure
- Comparison of SNAP-tag®/CLIP-tag™ Technologies to GFP
- Labeling with SNAP-tag® Technology Troubleshooting Guide
Feature Articles
Brochures
Selection Tools
Troubleshooting Guides
- Ke, N., Landgraf, D., Paulsson, J. and Berkmen, M. (2016) Visualization of Periplasmic and Cytoplasmic Proteins with a Self-Labeling Protein Tag. J Bacteriol; Jan 19;198(7), 1035-43. PubMedID: 26787765
- Clone and express once, then use with a variety of substrates
- Non-toxic to living cells
- Wide selection of fluorescent substrates
- Highly specific covalent labeling
- Simultaneous dual labeling
- Simultaneous dual protein labeling inside live cells
- Protein localization and translocation
- Pulse-chase experiments
- Receptor internalization studies
- Selective cell surface labeling
- Protein pull-down assays
- Protein detection in SDS-PAGE
- Flow cytometry
- High throughput binding assays in microtiter plates
- Biosensor interaction experiments
- FRET-based binding assays
- Single molecule labeling
- Super-resolution microscopy
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. All other trademarks are the property of their respective owners. The use of this product may require the buyer to obtain additional third-party intellectual property rights for certain applications. For more information, please email busdev@neb.com.
This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.