Applications of the Ph.D. Phage Display Peptide Libraries
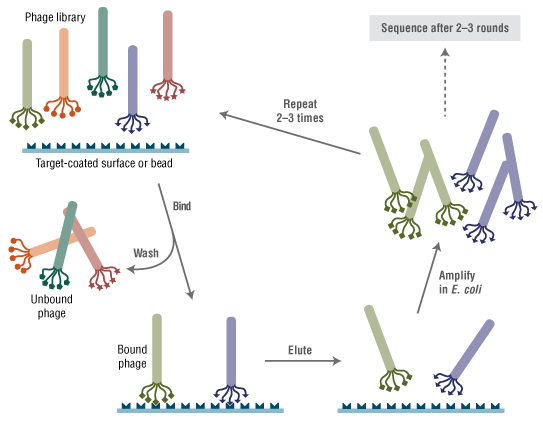
Phage display is a selection technique in which a library of peptide or protein variants is expressed as a genetic fusion to a bacteriophage coat protein. Each variant is displayed on the surface of a virion, while the DNA encoding the variant resides within the virion. This connection between genetic material and selectable function allows rapid identification of variants with desired properties by multiple rounds of affinity partitioning (panning) followed by phage amplification. Individual variants with the desired binding property can then be identified by simply sequencing the DNA contained within each selected phage. Combinatorial peptide libraries displayed on phage have allowed rapid screening of billions of different peptides against a wide variety of target molecules (reviewed in 1-3).
Over the last ten years, the Ph.D. libraries from New England Biolabs have become the dominant tools in this field, with hundreds of publications describing applications including epitope mapping (4-10), mapping protein-protein contacts (11-16) and identification of peptide mimics of non-peptide ligands (17,18). Bioactive peptides, which can be used as cell-targeting or gene delivery agents, have been identified either by panning against purified receptors (19-24) or against intact cells or tissue samples, both in vitro and in vivo (25-33). It is apparent that applications of the Ph.D. kits have been limited only by the imagination of the scientific community.
Anti-angiogenic therapy is being investigated as a cancer treatment strategy. VEGF is an important angiogenic factor, regulated by kinase domain receptor KDR/Flk-1, which is involved in both physiological and pathological pathways. Hetian and co-workers (21) panned against recombinant KDR (sKDR) to screen for peptides that would bind to KDR and potentially block its interaction with VEGF. A selected peptide sequence was found to inhibit proliferation of primary human vein endothelial cells, to have antiangiogenic effects in chick embryos, and finally to reduce breast tumor growth and metastasis by 70% in mice. Blocking protein-protein interaction with peptides discovered by phage display will have broad clinical applications for treatment of metastasizing tumors.
In order to elucidate the physiological function of the cell surface glycosoaminoglycan hyaluronan (HA), Mummert and co-workers (32) panned the Ph.D.-12 library against purified HA. The predominant selected peptide (Pep1) was determined to bind both free HA and HA expressed on cell surfaces, as well as to block HA binding to cell surfaces expressing the HA receptor, CD44. The activity of Pep1 facilitated studies asserting that HA is active in the trafficking of leukocytes at inflamed tissues. Highlighting the exceptional diversity of the Ph.D. libraries, neither alanine scanning nor scrambling of the Pep1 sequence resulted in clones with better activity than Pep1. Amazingly, when applied to skin, Pep1 prevented development of contact dermatitis upon exposure to a known allergen, underscoring connections between HA and immune responses to skin irritants, with broad clinical implications.
Genetic manipulation of dendritic cells (DC) could significantly improve our ability to control immune responses. Chamarthy and co-workers (25) identified a peptide by whole-cell panning, which is able to recognize murine DCs and function as a gene delivery vehicle when conjugated to a short DNA-binding domain. Up to 25% of DCs could be transformed using this construct when appended to polymeric microspheres.
in vivo panning
By applying Ph.D.-C7C library phage to the skin of a live animal and harvesting self-transfecting phage from the circulating blood, Chen and co-workers (34) identified a peptide that mediates protein delivery through skin. The predominant phage from blood samples was used to design a cyclic synthetic peptide which delivered both insulin and human growth hormone across skin. Furthermore, assays with the full complement of alanine scanned derivatives of the peptide revealed that the originally selected sequence was the best performer, emphasizing the diversity of the library. This exciting work may lead to a new class of systemic drug delivery agents with broad application since there is no requirement for physical association of the peptide with the drug.
Using a combination of in vitro and in vivo panning, Böckmann and co-workers (35) identified a peptide that targets human thyroid cancer cells over lung cancer cells, human embryonic kidney cells and VH6 fibroblasts. The observed specific binding and internalization of the peptide are promising for adenoviral gene transfer into thyroid tumors. In related work using transgenic mice with thyroid cancer, a single phage clone injected into the animals showed a 2-4X higher titer from harvested tumor cells over other tissues like kidney, liver, lung and heart (36). One sequence appeared in both studies indicating that murine and human derived thyroid cancer tumors share characteristic surface markers.
Anti-viral/microbial peptides
In a search for molecules that will inhibit entry of HIV-1 into cells, Ferrer and Harrison (20) panned all three Ph.D. libraries against an HIV envelope glycoprotein, gp120. A peptide was identified which binds gp120 and inhibits binding to the host protein CD4. A synthetic peptide with this sequence was found to inhibit HIV infection in vitro, and represents a promising lead for further drug development (37).
A peptide discovered by Mourez and co-workers (38) provided the basis for a polyvalent inhibitor of anthrax toxin that was able to nearly eliminate toxicity of anthrax in rats. The Ph.D.-12 library was panned against immobilized heptameric PA domain of B. anthracis toxin, and peptides specific for inactive, unassembled PA were eliminated by washing with soluble monomeric PA. Peptides specific for the toxic, heptameric form of PA were eluted competitively with soluble PA heptamer. The resulting peptides were specific for the toxic heptameric form but bound weakly; affinity was improved by tetramerizing the peptides on a soluble scaffold.
Mapping protein-protein interactions
Phage display can be used as an alternative to conventional two-hybrid systems to study protein-protein interactions. Carter and co-workers (11) were able to map contacts between two proteins involved in iron transport in E. coli. Panning against one of the proteins, TonB, selected for a number of sequences that align with FhuA, an OM receptor known to bind TonB. By using both linear and constrained loop libraries, a number of binding and structural motifs were identified spanning two distinct regions of FhuA. Binding interactions were confirmed using peptide-MBP fusions generating using the pMal-pIII vector (NEB #N8101).
Mirror image phage display
Mirror image phage display is based on the idea that if phage-displayed peptides (natural L-symmetry) bind to a synthetic target with D-symmetry then it may be assumed the D-peptide analogs of the phage clones will bind to the natural target (L-symmetry). These D-peptides are more resistant to proteolysis in animal systems and thus would have improved bioavailability. Wiesehan and co-workers (39) used mirror image phage display to identify D-peptide ligands for the Aβ fibril plaques of Alzheimer’s disease (AD). The target peptide for panning, Aβ (1-42), was synthesized entirely with D-amino acids. The predominant selected peptide was subsequently synthesized with D-amino acids and was shown to bind Aβ with micromolar affinity. Furthermore, in brain tissue sections from AD patients the D-peptide was able to stain plaques of natural Aβ fibrils much like Congo red dye, but unlike the dye did not stain plaques taken from five other non-AD samples. This work may lead to methods that monitor the disease progression in vivo, a crucial step in developing successful treatments.
Material-specific peptides
Angela Belcher has had a long-standing interest in harnessing the methods by which nature makes unique materials and surfaces for nanomaterials development. Her lab has carried out truly pioneering applications of the Ph.D. libraries for the synthesis and assembly of nanosized components into micrometer scale materials with unique electronic and/or optical properties. An initial publication described peptides discovered from panning against specific semiconductor surfaces which specifically bound to different crystalline compositions (e.g. GaAs(100) and Si) (40). In ensuing work, phage particles were engineered to express Ph.D.-selected CdS and ZnS binding peptides along their length via fusion to the pVIII major coat protein, resulting in spontaneous assembly of phage-templated nanowires (41). A viral film assembled in this way was stable at room temperature for seven months and phage were even infectious, demonstrating the reversibility of assembly (42,43). In yet another application, Sinesky and Belcher used the Ph.D. phage display to find peptides which detect surface defects in crystals (44). Surface defects are "inseparable/diffuse targets" but are essential knowledge for determining the quality of a given material. Finally, in an astonishing application, gold-binding peptides selected from the Ph.D.-12 library were used as the basis for a lithium ion battery electrode consisting of phage-templated gold-cobalt nanowires (45).
Small molecule binding peptides
Paclitaxel (Taxol) is an anti-mitotic agent. To elucidate in vivo protein factors with which taxol may interact, biotinylated paclitaxel was screened against a Ph.D. library (46). Comparison of the selected sequences with protein databases pointed to a human anti-apoptotic protein, Bcl-2 as the natural binding partner of paclitaxil. ELISA and circular dichroism experiments confirmed that paclitaxel binds to purified Bcl-2, indicating that phage display peptide libraries can in fact be used to discover biologically relevant peptide ligands for small molecules.
Substrate phage
The Ph.D. libraries can also be used to easily and rapidly determine the substrate specificity of enzymes that modify proteins by panning against an affinity resin specific for the modification. Sugimura et al. (47) used this method to determine the substrate specificity of mammalian transglutaminases (TGase), which carry out cross-linking reactions between glutamine residues and primary amines. The library was incubated with enzyme and a substrate amine (which included a biotin moiety), followed by capture of modified phage on monomeric avidin. Biotin-elutable phage presumably displayed a substrate peptide sequence for the enzyme. Consensus substrate sequences could be rapidly identified for a number of different TGases using this method.
A growing list of literature references for applications of the Ph.D. libraries is available here. If you know of a reference that is not included on this list, please contact us at phd@neb.com.
References
- Kehoe, J.W. and Kay, B.K. (2005) Chem. Rev. 105, 4056-4072.
- Brissette, R. et al. (2006) Curr. Opin. Drug. Discov. Devel. 9, 363-369.
- Paschke, M. (2006) Appl. Microbiol. Biotechnol. 70, 2-11.
- Eshagi, M. et al. (2006) Mol. Immunol. 43, 268-278.
- Davies, J.M. et al. (1999) Mol. Immunol. 36, 659-667.
- Gazarian, T.G. et al. (2003) Comb. Chem. High Throughput Screen. 6, 119-132.
- Osman, A.A. et al. (2001) Eur. J. Gastroenterol. Hepatol.13, 1189-1193.
- Santamaria, H. et al. (2001) Clin. Immunol.101, 296-302.
- Spillner, E. et al. (2003) Anal Biochem. 321, 96-104.
- Youn, J.H. et al. (2004) FEMS Immunol. Med. Microbiol. 41, 51-57.
- Carter, D.M. et al. (2006) J. Mol. Biol. 357,236-251.
- Bitto, E. and McKay, D.B. (2003) J. Biol. Chem. 278, 49316-49322.
- BouHamdan, M. et al. (1998) J. Biol. Chem. 273, 8009-8016.
- Dintilhac, A. and Bernues, J. (2002) J. Biol. Chem. 277, 7021-7028.
- Kenny, C.H. et al. (2003) Anal. Biochem. 323, 224-233.
- Wang, X.G. et al. (2004) J. Bacteriol. 186, 51-60.
- Hou, Y. and Gu, X.X. (2003) J. Immunol. 170, 4373-4379.
- Jouault, T. et al. (2001) Glycobiology, 11, 693-701.
- Du, B. et al. (2006) Biochem. Biophys. Res. Commun. 342, 956-962.
- Ferrer, M. and Harrison, S.C. (1999) J. Virol. 73, 5795-5802.
- Hetian, L. et al. (2002) J. Biol. Chem. 277, 43137-43142.
- Koolpe, M. et al. (2002) J. Biol. Chem. 277, 46974-46979.
- Luo, X. et al. (2002) Mol. Cell. 9, 59-71.
- Magdesian, M.H. et al. (2005) J. Biol. Chem. 280, 31085-31090.
- Chamarthy, S.P. et al. (2004) Mol. Immunol. 41, 741-749.
- Federici T (2006) J. Drug Targetting, 14, 263-271.
- Gazouli, M. et al. (2002) J. Pharmacol. Exp. Ther. 303, 627-632.
- Kragler, F. et al. (2000) EMBO J. 19, 2856-2868.
- Messmer, B.T. et al. (2000) J. Mol. Biol. 296, 821-832.
- Stratman, J. et al (2004) Infect. Immun. 72, 1265-1274.
- Curiel, T.J. et al. (2004) J. Immunol. 172, 7425-7431.
- Mummert, M.E. et al. (2000) J. Exp. Med. 192, 769-779.
- Nicklin, S.A. et al. (2004) J. Gene Med. 6, 300-308.
- Chen, Y. et al (2006) Nat. Biotechnol. 24, 455-460.
- Böckmann, M. et al (2005) J. Gene Medicine, 7, 179-188.
- Böckmann, M. et al (2005) Hum. Gene Ther. 16, 1267-1275.
- Biorn, A.C. et al (2004) Biochem. 43, 1928-1938.
- Mourez, M. et al. (2001) Nat. Biotechnol. 19, 958-961.
- Wiesehan, K. (2003) ChemBioChem, 4, 748-749.
- Whaley S.R. et al (2000) Nature, 405, 665-668.
- Mao C. et al (2003) Proc. Natl. Acad. Sci. 100, 6946-6951.
- Lee, S.W. et al (2002) Science, 296, 892-895.
- Lee et al (2003) Langmuir 19, 1592-1598.
- Sinensky, A.K. and Belcher, A. M. (2006) Adv. Mat. 18, 991-996.
- Nam et al. (2006) Science, 312, 885-888.
- Rodi, D.J. et al. (1999) J. Mol. Biol. 285, 197-203.
- Sugimura, Y. et al (2006) J. Biol. Chem. 281, 17699-17706.

