Competent Cells
Choose Type:
- Can I store competent cells at -20°C instead of -80°C?
- Does New England Biolabs offer a methyltransferase free strain of Competent E.coli?
- Does New England Biolabs offer strains of Competent E.coli suited for protein expression?
- What are the advantages of New England Biolabs' competent cells?
- What does the growth curve of NEB Turbo cells look like compared to DH5α?
- What is LysY?
- What volume of DNA can be added into competent cells?
- Which kind of transformation tubes should be used?
- What applications are SHuffle® strains useful for?
- Why did Synthetic Biologist Chris Voigt of MIT choose NEB 10-beta for DNA assembly and cloning?
- What formats are competent cells available in?
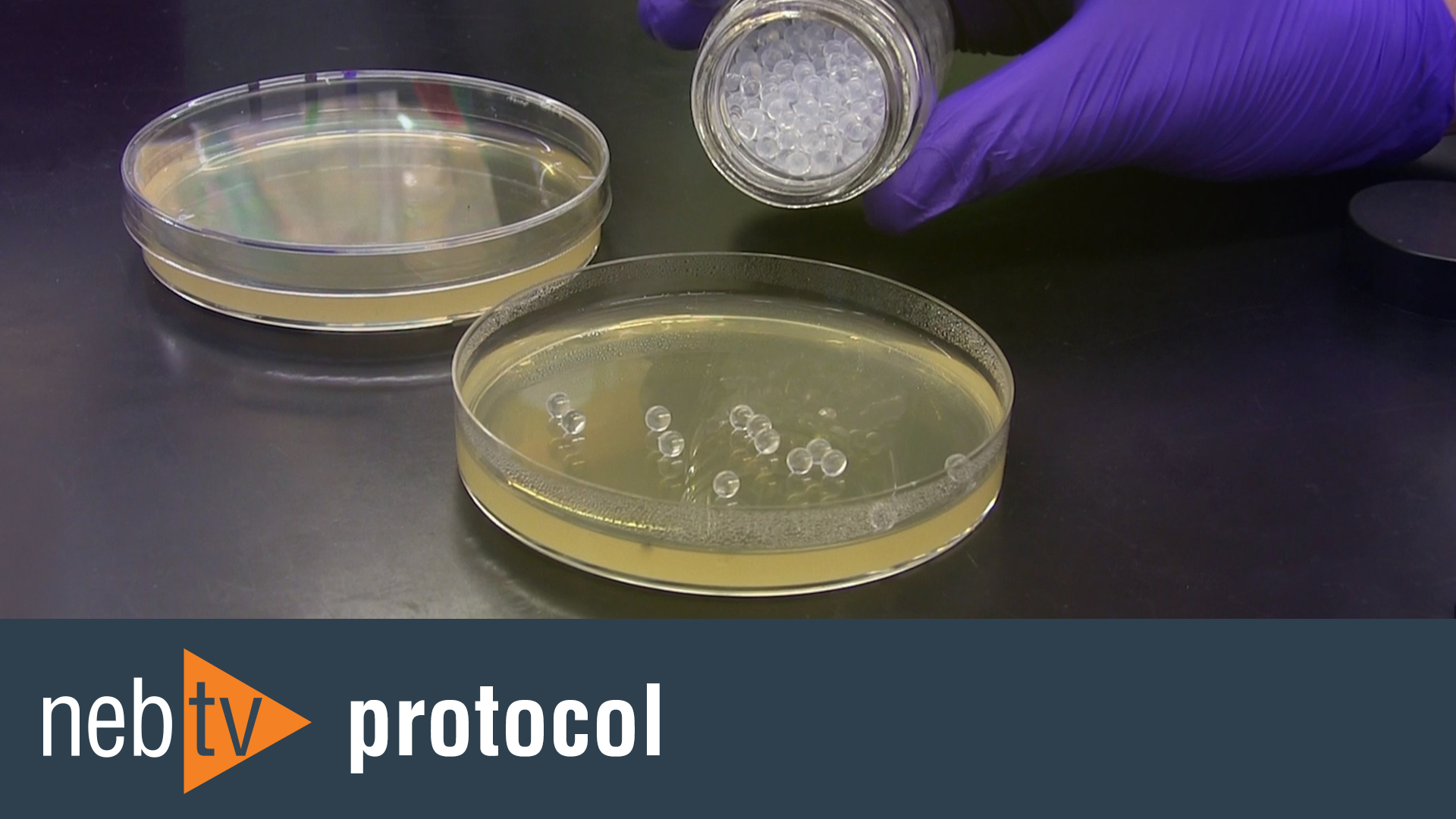
- Which strain of Competent E.coli should I use for general cloning?
-
Avoid Common Obstacles in Protein Expression
Read how to avoid common obstacles in protein expression that prevent interactions with cellular machinery.
- Competent Cell Brochure
- Protein Expression & Purification Brochure
- Characteristics of Select E.coli Strains
- Competent Cell Product Comparison
- Competent Cell Selection Guide
- Troubleshooting Transformation Reactions
- Additional E. coli Strain Genotypes
- Chemical Transformation Tips
- Competent E. coli Genome Sequences Tool
- Electroporation Tips
- Genetic Markers
- Making Unmethylated (Dam- Dcm-) DNA
- McrA, McrBC and EcoKI Strain Phenotypes
- Restriction of Foreign DNA by E. coli K-12
Feature Articles
Brochures
Selection Tools
Troubleshooting Guides
Usage Guidelines
- Schlegel, S., Klepsch, M., Gialama, D., Wickström, D., Slotboom, D.J. and de Gier, J. (2010) Revolutionizing membrane protein overexpression in bacteria Microb Biotechnol; 3 , 403-411 .
- Narayanan, A., Ridilla, M. and Yernool, D.A. (2010) Restrained expression, a method to overproduce toxic membrane proteins by exploiting operator–repressor interactions Protein Sci; 20 , 51-61 .
- Reddy, P.T., Brinson, R.G., Hoopes, J.T., McClung, C., Ke, N., Kashi, L. (2018) Platform development for expression and purification of stable isotope labeled monoclonal antibodies in Escherichia coli. mAbs MAbs; 10 (7), 992-1002. PubMedID: 30060704, DOI: 10.1080/19420862.2018.1496879
- Wagner, S., Klepsch, M., Schlegel, S., Appel, A., Draheim, R., Tarry, M., Hö, M., van Wijk, K.J., Slotboom, D.J., Persson, J.O. and de Gier, J. (2008) Tuning Escherichia coli for membrane protein overexpression Proc Natl Acad Sci U S A; 105 , 14371-14376 .
- Hibender, S. Landeta, C., Berkmen, M., Beckwith, J., Boyd, D. (2017) Aeropyrum pernix membrane protein VKOR promotes protein disulfide bond formation in two subcellular compartments. Microbiology; 163 (12), 1864-1869. PubMedID: 291309344
- Ren, G., Ke, N. and Berkmen, M. (2016) Use of the Shuffle Strains in Production of Proteins. Curr Protoc Protein Sci; Aug 1, 1;85:5.26.1-5.26.21.. PubMedID: 27479507 , DOI: 10.1002/cpps.11.
- Agrawal, A., Bisharyan, Y., Papoyan, A, Bednenko, J., Cardarelli, J., Yao, M., Clark, T., Berkmen, M., Ke, N., Colussi, P. (2019) Fusion to Tetrahymena thermophila granule lattice protein 1 confers solubility to sexual stage malaria antigens in Escherichia coli. Protein Expr Purif; 153, 7-17. PubMedID: 30081196, DOI: 10.1016/j.pep.2018.08.001.
- Leith, E.M., O'Dell, W.B., Ke, N., McClung, C., Berkmen, M., Bergonzo, C., Brinson, R.G., Kelman, Z (2019) Characterization of the internal translation initiation region in monoclonal antibodies expressed in Escherichia coli J Biol Chem; 294(48), 18046-18056.. PubMedID: 31604819, DOI: 10.1074/jbc.RA119.011008
- Reuter, W.H., Masuch, T., Ke, N., Lenon, M., Radzinski, M., Van Loi, V., Ren, G., Riggs, P., Antelmann, H., Reichmann, D., Leichert, L.I., Berkmen, M (2019) Utilizing redox-sensitive GFP fusions to detect in vivo redox changes in a genetically engineered prokaryote Redox Biol; 26, 101280. PubMedID: 31450103, DOI: 10.1016/j.redox.2019.101280
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. The use of this product may require the buyer to obtain additional third-party intellectual property rights for certain applications. For more information, please email busdev@neb.com.
This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.