NEB® 10-beta Electrocompetent E. coli
Supplied with outgrowth medium optimized for NEB 10-beta & NEB Stable Competent E.coli ; please do not use SOC outgrowth medium.
NEB 10-beta Competent E. coli is a derivative of the popular DH10B. It is T1 phage resistant and endonuclease I (endA1) deficient for high- quality plasmid preparations.
- High efficiency strain ideal for cloning large plasmids and BACs
- Available in chemically-competent format (single-use and 200 μl vials)
- No dry ice surcharge on competent cell shipments
- Outgrowth medium included
- Free of animal products
- Refer to our Electroporation Tips
Featured Video
-
Product Information
NEB 10-beta electrocompetent E. coli cells are optimized for high efficiency transformation by electroporation. These cells are ideal for DNA library constructions and all cloning purposes.
Highlights
- DH10B™ derivative
- Accomodation of large plasmids including BAC and cosmid constructs
- Efficient transformation of methylated DNA derived from eukaryotic sources and unmethylated DNA derived from PCR, cDNA and other sources
- Activity of nonspecific endonuclease I eliminated for highest quality plasmid preparations (endA1)
- Suitable for blue/white screening by α-complementation of the β-galactosidase gene (Φ80ΔlacZM15)
- Reduced recombination of cloned DNA (recA)
- Resistance to phage T1 (fhuA)
- K-12 Strain
- Free of animal products
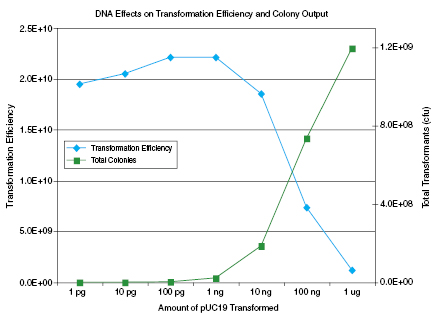
Transformation Efficiency
> 2 x 1010 cfu/μg pUC19Genotype
Δ(ara-leu) 7697 araD139 fhuA ΔlacX74 galK16 galE15 e14- Φ80dlacZΔM15 recA1 relA1 endA1 nupG rpsL (StrR) rph spoT1 Δ(mrr-hsdRMS-mcrBC)
- This product is related to the following categories:
- Cloning Competent Cell Strains Products
- This product can be used in the following applications:
- Transformation
-
Protocols, Manuals & Usage
-
Tools & Resources
-
FAQs & Troubleshooting
-
Citations & Technical Literature
-
Quality, Safety & Legal
Other Products You May Be Interested In
Ineligible item added to cart
Based on your Freezer Program type, you are trying to add a product to your cart that is either not allowed or not allowed with the existing contents of your cart. Please review and update your order accordingly If you have any questions, please contact Customer Service at freezers@neb.com or 1-800-632-5227 x 8.