Loop-mediated isothermal amplification allows testing to be performed anywhere
Posted on Tuesday, September 8, 2020
By
Topic: What is Trending in Science
Loop-mediated isothermal amplification (LAMP) is an emerging technology with a wide range of applications.
The polymerase chain reaction (PCR) is an indispensable tool in many molecular biology laboratories and has been transforming research and medical diagnoses for decades. Over the years, this technology has seen many advances. In the early days before thermocyclers became available, researchers sat diligently by multiple water baths set at different temperatures, timer in hand, ready to swap the tubes from one water bath to the next for each temperature-specific step – denaturation, annealing and extension. Before the use of heated thermocycler lids, mineral oil was layered on top of the reaction mixture to prevent evaporation. Then, Taq DNA Polymerase (NEB #M0273), the first heat-stable polymerase, was introduced. Prior to this discovery, a heat-labile polymerase was laboriously added at the start of every cycle because it was destroyed by the high temperatures required for the denaturation step. Scientists could now add polymerase for the entire amplification at the beginning of the experiment. Reaction set up became even more user-friendly with the introduction of hot start polymerases, in which dissociation from an inhibitor is required for activity, allowing for increased specificity and room temperature reaction set up.
Now, PCR is much more efficient and a mainstream laboratory technique for a wide range of applications. It requires equipment such as a thermocycler and gel electrophoresis or real-time instrumentation, which require an electricity source. These are readily available in well-equipped laboratories – along with the knowledge required to design and carry out a PCR. However, in some instances there are time-sensitive, or location-specific aspects to a study that make transport of samples to a laboratory the limiting factor in acquiring prompt, accurate results. In these cases, the need for a simpler form of nucleic acid amplification became apparent – not in place of traditional PCR, but as an option for use in a broader range of settings.
The speed and simplicity of LAMP
In response to this need, researchers developed a new method of nucleic acid amplification: loop-mediated isothermal amplification (LAMP). In a nutshell, LAMP is a powerful method for amplifying a target sequence in a single tube, at a single temperature using a variety of simple detection methods.
LAMP uses 4 to 6 primers that recognize 6 to 8 regions of the target DNA or RNA sequence. The design of two of the primers intentionally results in self-hybridizing loop structures that form a dumbbell, providing numerous sites for synthesis initiation. Bst DNA Polymerase, Large Fragment (NEB #M0275), which is active at elevated temperatures and ideal for this application, displaces downstream DNA that is encountered during strand synthesis. These features of LAMP – the hyper priming of the target sequence, coupled with the use of a strand-displacing DNA polymerase – lead to exponential amplification at a rate that can be detected by a variety of methods in approximately 30 minutes.
Here's how it works in more detail:

NEB-engineered versions of the Bst enzyme have expanded LAMP utility by reducing inhibition by common sample contaminants and dUTP. Increased dUTP tolerance enabled carryover prevention, once common only in PCR-based techniques, to be adapted to LAMP. Additionally, robust RNA-based amplification can be accomplished by the simple addition of the NEB WarmStart RTx enzyme, which is also included in NEB LAMP master mixes. Therefore, amplification of RNA or DNA substrates can be performed outside the walls of a laboratory; in fact, the entire reaction takes place at 65°C and could be carried out in a cup of hot water.
One of the advantages of LAMP is its broad utility. LAMP reaction progress can be tracked in real time by adding a fluorescent dye that binds to dsDNA as it amplifies. For high throughput applications, LAMP can be incorporated into automated workflows, where endpoint detection is measured via an absorbance plate reader. It can also be adapted for performance outside of a traditional lab, where analysis “in the field” requires a simplified output that shows amplification success. Various detection methods have been tested, including visualizing the precipitation of magnesium pyrophosphate or the color change of a metal-sensitive indicator, but both of these methods are difficult to detect by eye and require approximately 60 minutes before detection is possible.
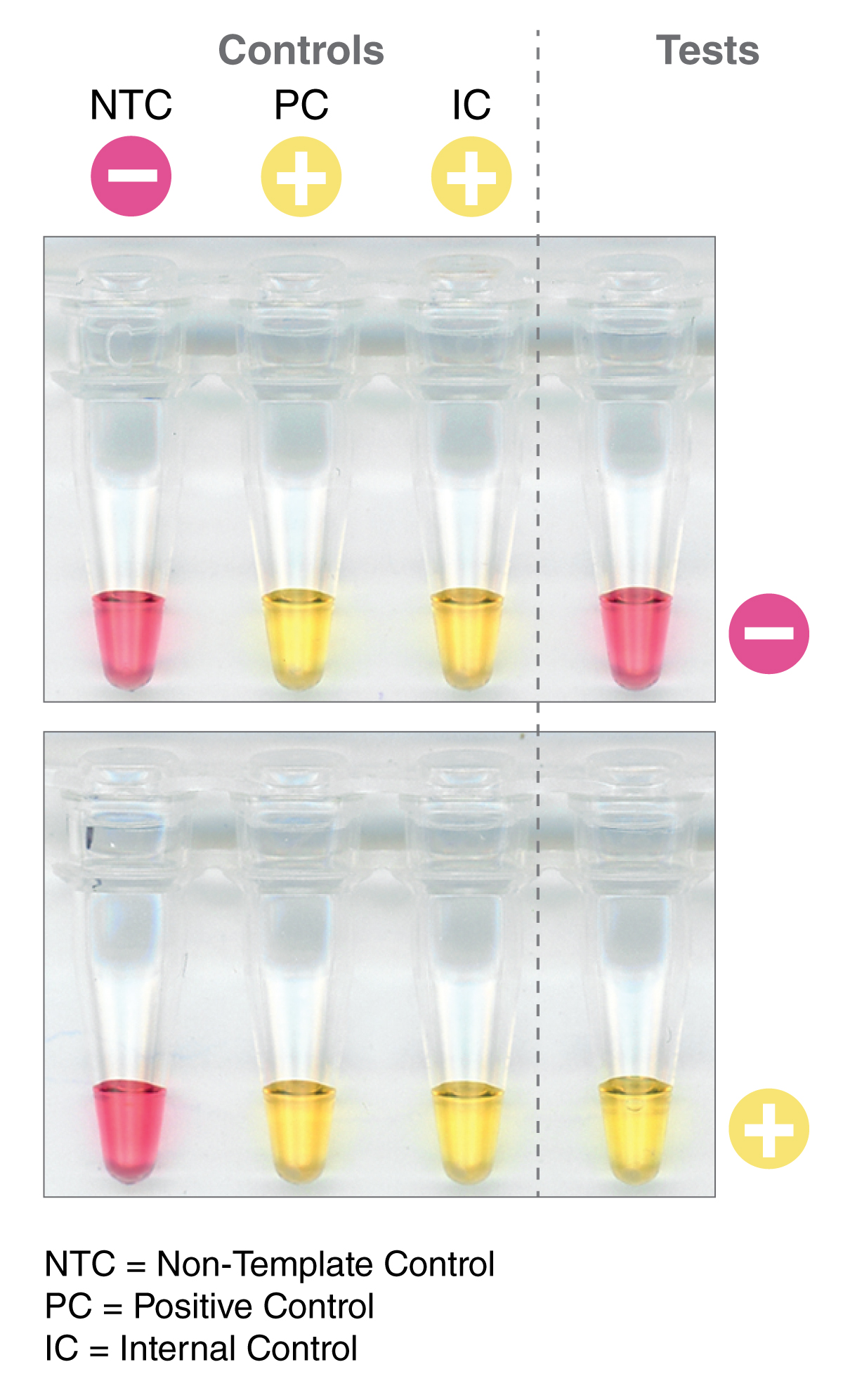
NEB scientists developed a solution to this - a very visible, pH-based colorimetric detection. This method utilizes a pink-to-yellow color change that is visible to the naked eye, resulting from a change in pH. During amplification, a proton is released with every dNTP that is incorporated into the growing DNA strand; in the presence of a lightly buffered solution, this leads to a decrease in pH of 2-3 units, which is attributed to the large amount of DNA made in LAMP reactions. Incorporation of a pH indicator directly into the reaction mixture enables visual detection of amplification. This eliminates the need for specialized detection methods, such as gel electrophoresis, which is not practical in remote settings where point-of-care diagnostics might be desirable or for high throughput analysis in field studies. As a result, LAMP lends itself well to a variety of applications.
LAMP IN THE FIELD - Eradicating dengue
There is no better example of just how beneficial amplification in the field is than in the work conducted by the World Mosquito Program (WMP) (worldmosquitoprogram.org), whose research aims to eradicate vector-borne illnesses.
Dengue, chikungunya and Zika viruses are transmitted by arthropods that acquire the diseases by feeding on infected human blood and subsequently passing it to the next human they bite. The mosquito (Aedes aegypti), is one of these arboviral disease-disseminating organisms. The World Health Organization estimates that 2.5 billion people live in dengue transmission areas; it is one of the top ten global health threats and the most rapidly spreading mosquito-borne disease.
Interestingly, 40-60% of all the different species of insects worldwide (including butterflies and dragonflies) contain a maternally-transmitted, endosymbiotic Gram-negative bacterium called Wolbachia pipientis in their reproductive cells; Aedes aegypti mosquitoes are one of the few insects that do not normally carry Wolbachia. Once a virus (e.g., dengue) enters cells, the presence of Wolbachia in those cells prevents viral growth by slowing replication and rapidly degrading viral RNA, preventing downstream infections in humans.
This potential biocontrol strategy was recognized by the WMP and researchers embarked on a massive effort to stop transmission by disseminating Wolbachia-containing mosquitoes in dengue- endemic countries in Central and South America and into Asia and the Pacific regions. Researchers injected the wMel strain of Wolbachia from fruit flies into Aedes aegypti eggs and the Wolbachia-infected mosquitoes were released in Northern Australia on a controlled schedule. Within a few months, close to 100% of mosquitoes contained Wolbachia, an infection rate that has been retained years later.
The WMP adapted LAMP for field analysis and compared it with the established TaqMan® qPCR assay. LAMP primers were designed to detect the wsp gene from two Wolbachia strains. Using the WarmStart® Colorimetric LAMP 2X Master Mix (NEB #M1800), primers and target DNA they found that in just 30 minutes at 65°C, a simple color change can reliably determine if the mosquitoes are infected with Wolbachia: pink = negative (mosquito does not contain Wolbachia DNA), yellow = positive (mosquito contains Wolbachia DNA).
Screening mosquitoes in the field using colorimetric LAMP is inexpensive compared to the TaqMan qPCR assay, and it avoids the lengthy process of transporting samples back to a specialized laboratory for analysis. Results are in real time, and this improves accuracy regarding the geographical localization of the Wolbachia-containing mosquitoes.
LAMP IN SPACE – Monitoring astronaut genomes
The field of space biology is still in its infancy, but an ever-expanding list of biological experiments is being conducted on the International Space Station (ISS) each year, some with the help of students, who design these experiments as part of the Genes in Space contest (www.genesinspace.org). In early experiments, reactions to be conducted on the ISS were typically prepared on Earth before being executed in space, and then transported back to Earth where they could be analyzed and interpreted. In this way, the first PCR was conducted in space in 2016 aboard the ISS. But with the goal of extended space travel there remained a need to enable astronauts to analyze reactions that could be conducted on station without having to send samples back down to Earth. In March 2017, colorimetric LAMP experiments were conducted on the ISS to detect a specific repetitive telomeric DNA sequence and observe the colorimetric results. Telomeres are DNA-protein structures at the ends of chromosomes that support chromosomal stability by protecting them from degradation; abnormal telomere shortening is associated with human disease. The colorimetric LAMP assays that were designed to assess telomere dynamics were prepared on the ground but after amplification on station using a portable miniPCRTM instrument, the results (a simple pink to yellow color change enabled by the WarmStart Colorimetric LAMP Master Mix) were easily visualized, indicating successful amplification. Traditional PCR using both Taq and Q5® DNA polymerases (NEB #M0494) were run in parallel and all reactions were compared to those run on Earth to demonstrate the study’s success. Analysis of the LAMP results on the ISS was made possible by the fact that colorimetric LAMP required only visual inspection – photographing the tubes against a white piece of paper highlighted the simplicity of the methodology and its suitability for even one of the most extreme settings.
LAMP DURING A PANDEMIC – Developing LAMP for COVID-19 screening
During a global pandemic, rapid, widespread testing of the infectious agent is critical – not just for testing symptomatic cases, but also as a screening method as people return to work, and to generally monitor infection levels in a community.
The SARS-CoV-2 virus causes symptoms that resemble the flu, so molecular diagnosis is essential for confirmation of infection and epidemiological monitoring. RT-qPCR is the standard for the diagnosis of acute infections from upper and lower respiratory tract specimens, and testing is typically conducted in authorized labs that can perform high complexity tests. However, the scientific community is collaborating to develop faster, alternative approaches to detect this pathogen.
Scientists at NEB demonstrated the ability of a colorimetric LAMP assay to detect RNA from the SARS-CoV-2 virus that causes COVID-19. The assay was evaluated using samples from respiratory swabs from confirmed infections from patients in Wuhan, China (Figure 1) where isothermal amplification of SARS-CoV-2 RNA leads to a color change from pink to yellow (Figure 2). Additional studies have examined colorimetric LAMP in comparison to commercial RT-qPCR tests. LAMP can be used with samples that do not require RNA isolation, including those using human saliva as a sample input. NEB recently released a research-use only (RUO) product based on this simple technique, the SARS CoV-2 Rapid Colorimetric LAMP Assay Kit (NEB #E2019). For more details, see page 5. Colorimetric LAMP has huge potential as a disease screening method during a pandemic because of the low cost, rapid results and ease of use in a broad range of settings. The quick sample preparation and portability of this assay represent the next-generation of point-of-need diagnostics that will aid in disease prevention or containment and lead to improved outcomes.
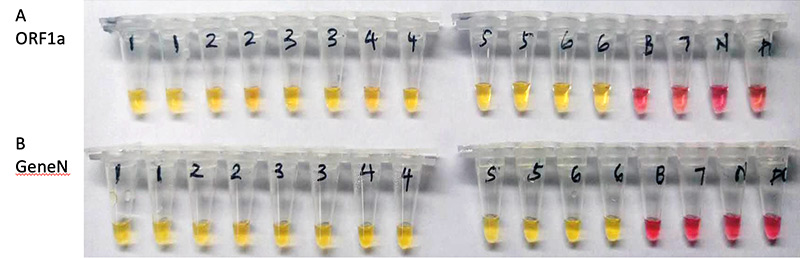
Figure 1: SARS-CoV-2 detection from COVID-19 patient samples in Wuhan, China. Samples testing positive (1-6) or negative (7) with commercial RT-qPCR tests were assayed using colorimetric LAMP assay with primer set targeting ORF1a (A) and GeneN (B). Yellow indicates a positive detection after 30 min incubation, and pink a negative reaction with results compared to the negative control (N). B, Blank control without template. P, samples containing a plasmid used as positive control for qPCR.
LAMP IN AGRICULTURE – Protecting crops and saving money
The widespread use of colorimetric LAMP doesn’t end there. Due to its sensitivity and low cost (under a dollar per sample) it is also being applied in agriculture. For example, Grapevine red blotch virus (GRBV) is spread by the three-cornered alfalfa leafhopper and it affects the viticulture industry in North America. It causes a reduction in both yield and quality, and while characteristic leaf changes are observed, they are not reliable and so diagnostic testing is required. The sample collection is as simple as inserting a sterile pipette into the leaf petiole and then incubating the tip in sterile water. Sample testing using a colorimetric LAMP assay takes place on site. Compare this with the time and cost of traditional methods for screening for GRBV – sample collection, transport to a specialist lab, a 2-hour DNA extraction method and PCR analysis. Additionally, the sensitivity of the LAMP assay was reported to be 2 orders of magnitude lower than traditional PCR and qPCR methods.
The value of colorimetric LAMP lies in its simple and inexpensive application in study settings that have not previously been PCR-friendly. Colorimetric LAMP opens up opportunities for human health and medical diagnosis that until recently, have not been possible due to the specialized requirements of traditional PCR. Any study environment whereby samples currently need to be transported from the field to a specialized lab is an opportunity for the development of a colorimetric LAMP assay.
NEB will not rent, sell or otherwise transfer your data to a third party for monetary consideration. See our Privacy Policy for details. View our Community Guidelines.
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. See www.neb.com/trademarks. The use of these products may require you to obtain additional third-party intellectual property rights for certain applications. For more information, please email busdev@neb.com.
Don’t miss out on our latest NEBinspired blog releases!
- Sign up to receive our e-newsletter
- Download your favorite feed reader and subscribe to our RSS feed
Be a part of NEBinspired! Submit your idea to have it featured in our blog.


