DNA Assembly and Cloning NEBuilder® HiFi DNA Assembly NEBuilder® HiFi DNA Assembly – Benefits Over GeneArt Gibson Assembly® and In-Fusion® Snap Assembly
NEBuilder® HiFi DNA Assembly – Benefits Over GeneArt Gibson Assembly® and In-Fusion® Snap Assembly
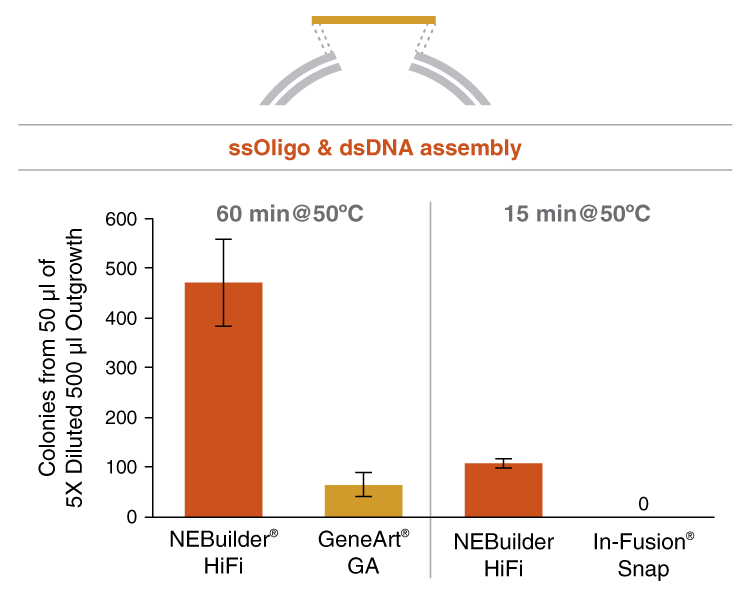
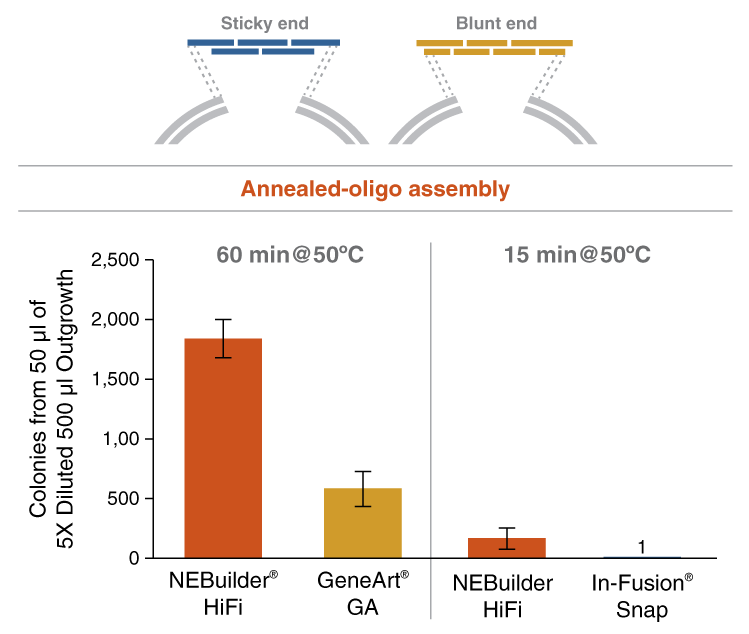
Return to NEBuilder® HiFi DNA AssemblyNEBuilder HiFi DNA Assembly offers several advantages over GeneArt Gibson Assembly and In-Fusion Snap Assembly. These include: higher accuracy due to the use of a high-fidelity polymerase, the ability to assemble both 5´- and 3´-end mismatches, lower DNA input requirements and the ability to bridge two dsDNA fragments with a ssDNA oligo. NEBuilder HiFi is the clear choice for efficient and accurate DNA assembly.
Performance Data
See how NEBuilder HiFi outperforms GeneArt Gibson Assembly and In-Fusion Snap Assembly
- Improved thermostability
- 2-Fragment assembly
- 4-Fragment assembly
- 6-Fragment assembly
- ssDNA oligo with linearized vector
- Annealed ssDNA oligos with 3´ and 5´ overhangs to a linearized vector
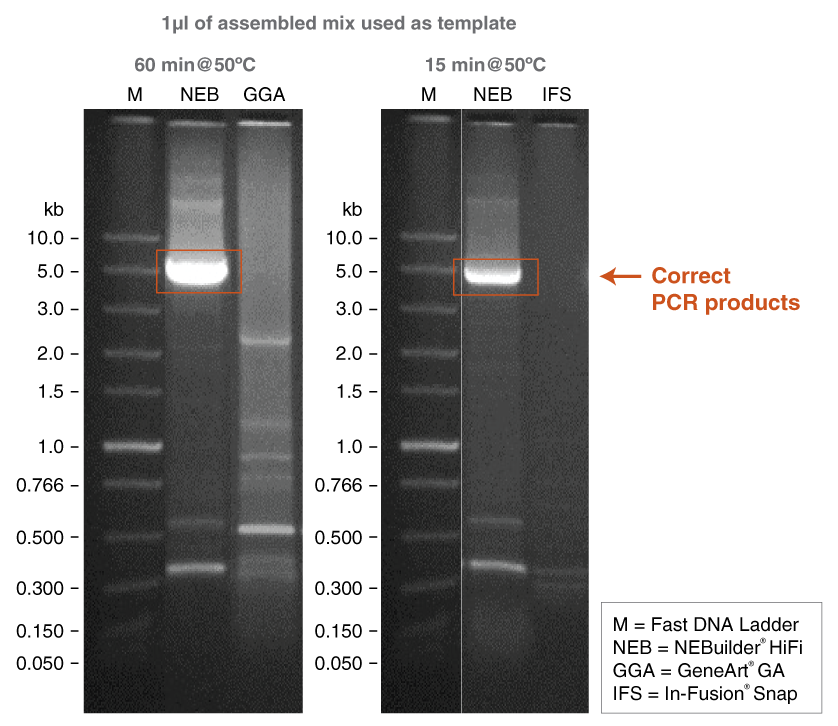
- Amplification of 12-fragment DNA assembly product
- In vitro enrichment of assembled DNA by phi-29 DNA Polymerase
Figure 1: NEBuilder HiFi DNA Assembly offers improved thermostability
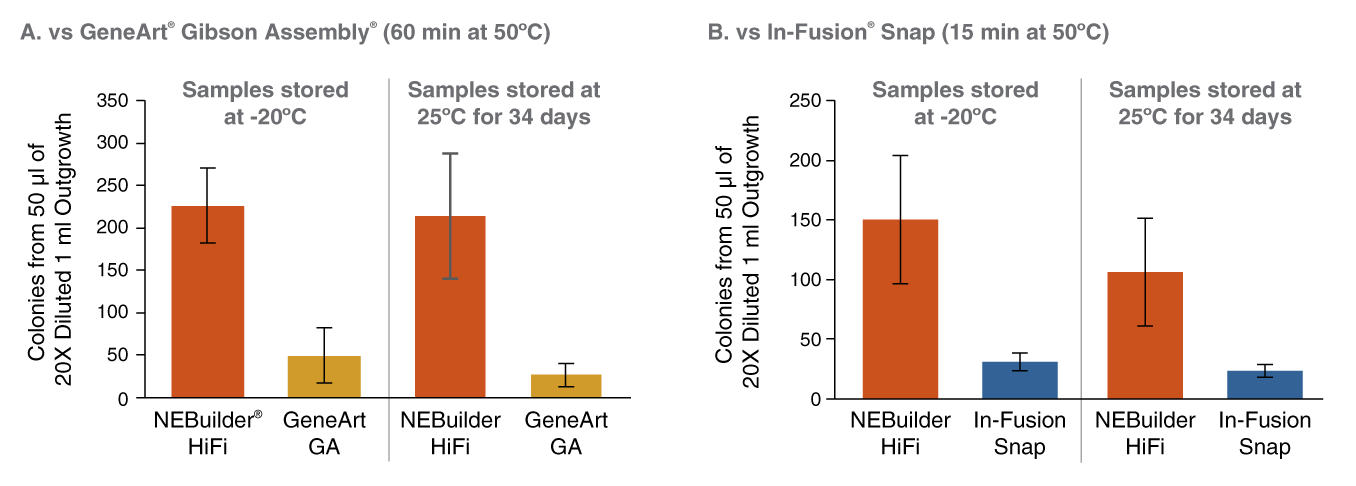
* Positive control from In-Fusion Snap Assembly Master Mix (Takara Bio USA)
Figure 2: NEBuilder HiFi DNA Assembly offers improved efficiency in 2-fragment assembly reactions
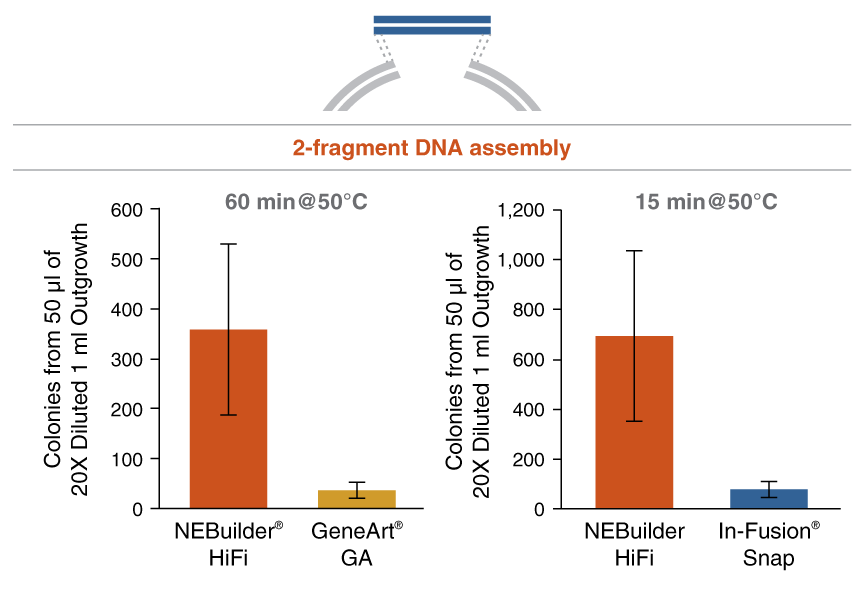
* Positive control from In-Fusion Snap Assembly Master Mix (Takara Bio USA)
Figure 3: NEBuilder HiFi DNA Assembly offers improved efficiency in 4-fragment assembly reactions
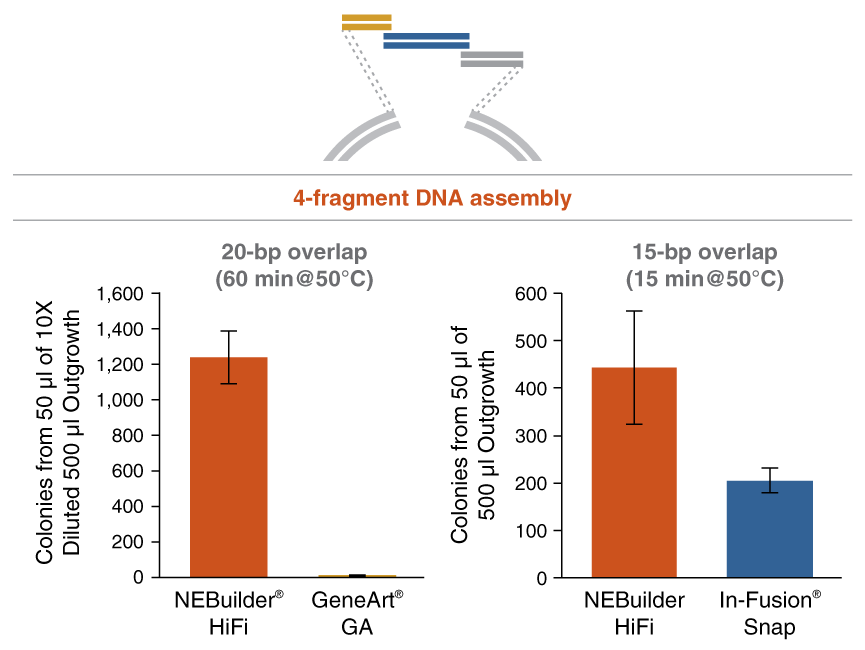
Figure 4: NEBuilder HiFi DNA Assembly offers improved efficiency in 6-fragment assembly reactions
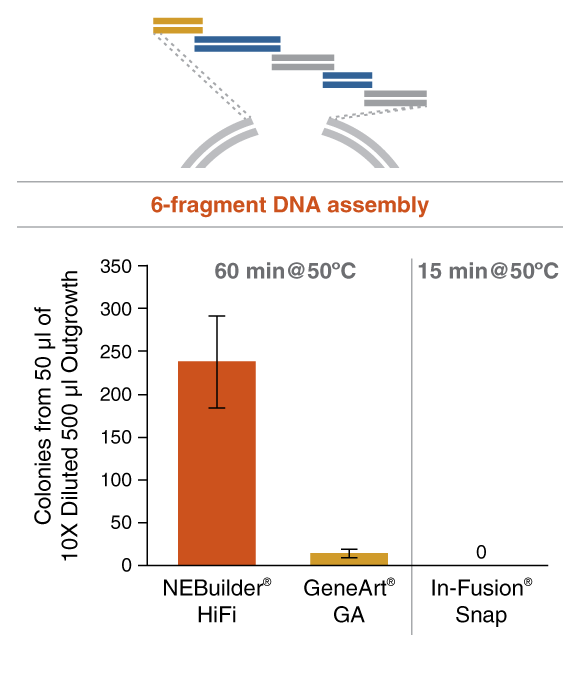
Figure 8: In vitro enrichment of 20 kb assembled DNA by phi-29 DNA Polymerase
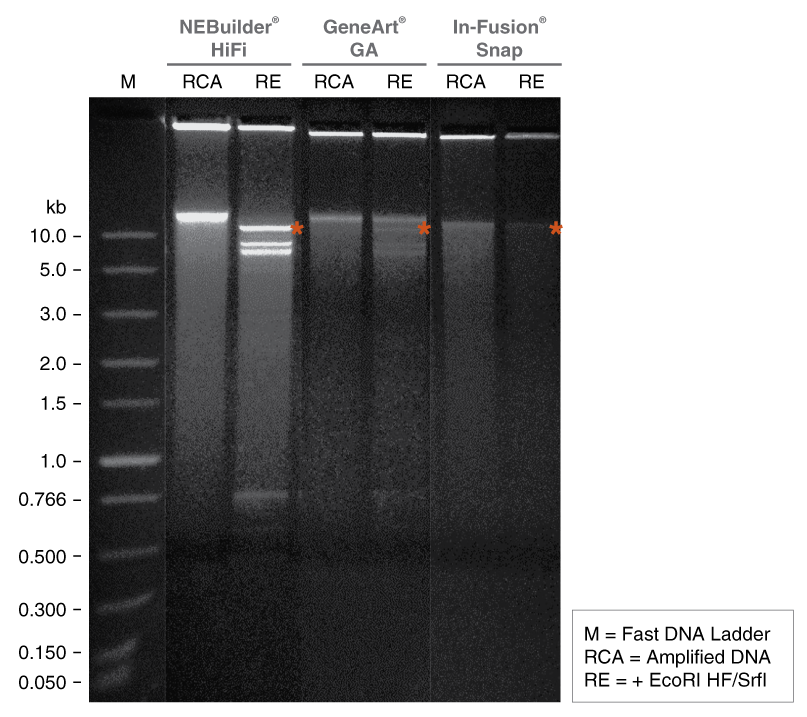
One or more of these products are covered by patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc. For more information, please email us at busdev@neb.com. The use of these products may require you to obtain additional third party intellectual property rights for certain applications.