Protocol for High Molecular Weight DNA (HMW DNA) Extraction from Tissue (NEB #T3060)
Below is a detailed protocol for the extraction of high molecular weight DNA from tissue samples. Please refer to the complete product manual for more detailed guidance. For help selecting input amounts, visit Choosing Input Amounts for the Monarch HMW DNA Extraction Kits. If you are using this kit upstream of ultra-long DNA sequencing on the Oxford Nanopores Technologies platform, please also review our Ultra-Long DNA Sequencing Workflow®’ Guidance for HMW DNA Extraction Upstream of Oxford Nanopore Technologies as well as our Protocol for UHMW DNA Cleanup in the Oxford Nanopore Technologies® UL Library Prep Workflow.
Materials Required but Not Supplied:
- Thermal mixer containing a 2 ml tube block (if not available, use a 1.5 ml block).
- Isopropanol, 550 µl per sample (Low Input: 275 µl/sample).
- Ethanol (≥ 95%).
- 1.5 ml DNase-free, low DNA binding microfuge tubes (e.g., Eppendorf® DNA LoBind®, #0030108051) are recommended for elution and storage (1 per sample); it is especially important to use low DNA binding tubes if working with UHMW DNA, which tends to bind to plastic surfaces.
- Recommended: vertical rotating mixer (e.g., Thermo Scientific® HulaMixer® Sample Mixer).
- Wide-bore pipette tips.
- Microcentrifuge
- Optional: rotor-stator homogenizer (e.g., TissueRuptor II), and compatible 2 ml tubes
- Optional: 5 M NaCl for liver samples
Important Notes Before You Begin:
- Review the complete protocol before beginning.
- Add ethanol (≥ 95%) to the gDNA Wash Buffer as indicated on the bottle label.
- Preheat thermal mixer with 1.5 ml block to 56°C.
- Proteinase K and RNase A should be stored at -20°C.
Starting Material Notes:
Use the table below to determine the designation of your sample type, which will determine various volumes in the protocol. If processing liver samples, follow specific guidance provided after Step 11 in Part 1.
|
PROTOCOL DESIGNATION |
TISSUE AMOUNT |
|---|---|
|
Standard Input |
10 – 25 mg for most tissues |
|
Low Input |
5 – < 10 mg* or |
- Fresh Tissue
– Ensure tissues are cut into the smallest possible pieces
– Pestle homogenization typically produces gDNA 50 kb to ≥ 500 kb
– No performance difference has been observed using a rotor-stator homogenizer
- Frozen Tissue
– Tissue powders are recommended over tissue pieces, as lysis is more efficient, and handling is easier
– Freshly or recently prepared frozen tissue powder samples provide better yields than those stored long term at -80°C
– If working with a powder is not possible, ensure frozen tissue is cut into the smallest possible pieces
– Rotor-stator homogenization has been shown to improve ligation-based nanopore sequencing by generating shorter gDNA fragments (50–250 kb)
– Samples homogenized using the pestle may benefit from needle shearing after elution. See guidance in “Needle Shearing” in the product manual.
HMW gDNA Purification Consists of Two Stages:
PART 2: HMW gDNA Binding and Elution
PART 1: Tissue Lysis
- Prepare a master mix of HMW gDNA Tissue Lysis Buffer and Proteinase K according to the table below and the number of samples that will be processed. There is no need to make excess master mix.
- Transfer the desired amount of tissue to a Monarch Pestle Tube (or, if using a rotor-stator homogenizer, to a compatible 2 ml tube, not included). Place on ice (fresh samples) or dry ice (frozen samples).
- Briefly spin the sample (2–3 seconds) in a benchtop minicentrifuge to collect all tissue material at the bottom of the tube. If working with frozen tissue powder and some of the powder is stuck to the tube, let the sample thaw briefly before spinning.
- Homogenization can be carried out using one of two methods: using a pestle or rotor-stator homogenizer. If using a rotor stator homogenizer, the sample must be in a 2 ml tube (not provided). If working with multiple samples, each sample should be taken quickly through all sub-steps and placed in the thermal mixer before processing the next sample. Homogenization initiates endogenous nuclease activity, making it essential to take each sample through to incubation before starting the next sample.
Homogenization Using Pestle
 If working with multiple samples, perform steps a-f for one sample and then repeat with the next sample.
If working with multiple samples, perform steps a-f for one sample and then repeat with the next sample.- For frozen samples, thaw at room temperature and quickly proceed to the next step.
- Use the pestle to grind the sample within the pestle tube; leave the pestle in the tube.
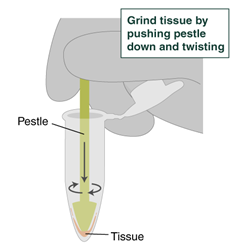
- Using a wide-bore pipette tip, add 600 µl (Low Input: 300 µl) of the lysis master mix to the sample. Do not dispose of this tip yet, as it will be used to mix the sample.
- Ensure there is no tissue material remaining on the pestle, then discard the pestle. If visible DNA threads or tissue material stick to the pestle, transfer it carefully into the tube by wiping the pestle tip along the tube rim. When working with brain tissue, some tissue material may accumulate at the rim of the pestle tip. Remove this material using the tip of a regular 200 µl pipette tip and transfer it back into the tube.
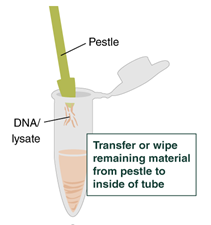
- Using the wide-bore tip, pipette up and down a few times to homogenize the tissue lysate and ensure rapid, complete lysis. Discard the pipette tip.
- Begin incubation in the thermal mixer (Step 5), and repeat steps a-f with any remaining samples.
- For frozen samples, thaw at room temperature and quickly proceed to the next step.
- For frozen samples, thaw at room temperature and quickly proceed to the next step.
- Add 600 µl (Low Input: 300 µl) of the lysis master mix to the sample. If sample is not in a 2 ml tube (not provided), transfer at this step.
- Ensure the tissue pieces can move freely by flicking or vortexing briefly.
- Insert the tip of the homogenizer probe into the sample and turn on to the lowest setting. Homogenize until tissue pieces are no longer visible. Suggested homogenization time is 5–20 seconds; stop when foam begins to form in the lysate. Additional homogenization may be required to reach optimal gDNA size for ONT sequencing (50–250 kb). gDNA size can be verified by pulsed field gel electrophoresis or FEMTO Pulse®. Rotor-stator homogenizers may run at higher speeds after extended use; reduce homogenization time if necessary.
- Transfer sample to a 1.5 ml Pestle Tube and begin incubation in the thermal mixer (Step 5). Exchange or clean the rotor-stator probe as recommended by the manufacturer and repeat steps a-e with remaining samples.
- Incubate samples in a thermal mixer at 56°C for a minimum of 45 minutes with agitation at the desired speed. When lysis is complete, samples will turn from turbid to clear (or mostly clear, depending on the tissue type). The speed of the thermal mixer influences fragment length and lysis; higher agitation speeds reduce DNA size and sample lysis time. For most applications, including the standard ligation-based Oxford Nanopore Technologies (ONT) sequencing protocols, maximum agitation speed (1400-2000 rpm) is recommended.
- Maximum agitation speed (1400–2000 rpm) will produce DNA fragments predominantly 50–250 kb and ranging up to ~500 kb.Under these agitation conditions, tissue samples will be rapidly digested and are easy to process.
- For maximum DNA length (“UHMW DNA”, up to Mb range for soft organ tissues), agitate at 500–700 rpm. During incubation, mix samples every 10–15 minutes (more frequently for DNA-rich samples) by inverting a few times to ensure complete lysis. With low agitation speeds, tissue samples are digested very slowly, and the isolated HMW gDNA is significantly “tangled”, reducing the efficiency of protein removal in later steps. This DNA is also difficult to dissolve during elution and can result in visible DNA aggregates.
- For highest yields, particularly when working with low input samples or samples with relatively low DNA content (e.g., brain and muscle), stop agitation after 15 minutes and continue the remaining incubation time without agitation.
- Add 10 µl (Low Input: 5 µl) RNase A and mix by inverting 5–10 times. Incubate for 10 minutes at 56°C at the same agitation speed used in Step 5. If working with brain tissue: after RNase A digestion, chill the lysate on ice for 3 minutes before adding the Protein Separation Solution to ensure efficient phase separation.
- Change the heat block in the thermal mixer to accommodate a 2 ml tube, and preheat the block to 56°C. If a 2 ml heat block is not available, continue working with the 1.5 ml block.
- Add 300 µl (Low Input: 150 µl) Protein Separation Solution and mix by inverting for 1 minute. Alternatively, a vertical rotating mixer at 20 rpm can be used.
- Centrifuge for 10 minutes at 16,000 x g. If working with multiple samples, during centrifugation, prepare the plastics for Part 2, as indicated in the following step. The sample will separate into a large, clear phase (DNA) and a smaller (often brown) protein phase, usually on the bottom of the tube. For some tissues, the protein phase may be yellow or clear. Additional centrifugation time (10–20 minutes) may be required for complete phase separation, particularly when low agitation speeds were used.
- If working with multiple samples, prepare and label the plastics for the upcoming steps. Each sample will require:
- 1 Monarch Collection Tube II (no need to label)
- 1 Monarch Bead Retainer inserted into the collection tube; this will be used to remove the wash buffer from the gDNA bound to the beads.
- 2 Monarch 2 ml Tubes; one for phase separation and one for elution.
- 1 1.5 ml microfuge tube (DNA low bind recommended, not provided); this will be used to collect the eluate.
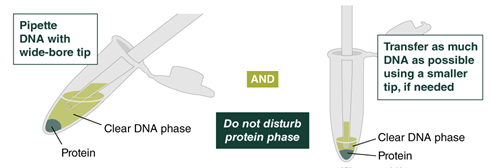
- Using a 1000 µl (Low Input: 200 µl) wide-bore pipette tip, transfer the upper phase containing the DNA (large, clear phase) to a labeled Monarch 2 ml Tube. A substantial fraction of HMW DNA will be located at the interface between the clear upper phase and the protein phase; highest yields will be achieved by transferring as much of the upper phase as possible. Using a 200 µl wide-bore pipette tip to transfer the final volume of the upper phase is recommended for maximum yield. Avoid transferring material from the protein layer, although a small amount (1–2 µl) will not be detrimental. If a small amount of the protein phase enters the pipette tip, gently push it back into the tube. If a lower protein phase is not visible, leave ~30 µl behind to ensure protein is not carried over. Typically, the transferred volume will be ~ 800 µl (Low Input: ~400 µl). If the volume of the sample is < 700 µl (Low Input: < 350 µl), adjust the volume of isopropanol used in Step 2 of Part 2: HMW gDNA Binding and Elution to 0.7 volumes.
 If processing liver samples, see guidance below in “Processing Liver Samples”.
If processing liver samples, see guidance below in “Processing Liver Samples”.
|
PROTOCOL DESIGNATION |
VOLUME OF HMW gDNA TISSUE LYSIS BUFFER PER SAMPLE (µl) |
VOLUME OF PROTEINASE K PER SAMPLE (µl) |
|---|---|---|
|
Standard Input |
600 |
20 |
|
LOW INPUT |
300 |
10 |
Homogenization Using Rotor-Stator Homogenizer
![]() If working with multiple samples, perform steps a-e for one sample and then repeat with the next sample.
If working with multiple samples, perform steps a-e for one sample and then repeat with the next sample.
Processing Liver Samples
Copurification of polysaccharides (i.e., glycogen) often results in variable or low A260/A230 values. Addition of NaCl to the sample before DNA capture helps to remove polysaccharides, resulting in A260/A230 values > 2.0. The following protocol modifications are recommended:
- Following Step 11 of Part 1 (phase separation): To 650 µl of the upper phase (Low Input: use all, ~400 µl), add 500 µl (Low Input: 315 µl) 5 M NaCl to reach a final concentration of 2.2 M NaCl. Pipette with a wide-bore tip or gently invert to mix.
- At Step 2 of Part 2, add 800 µl (Low Input: 500 µl) isopropanol (0.7 volumes of isopropanol per volume of upper phase + NaCl).
PART 2: HMW gDNA Binding and Elution
- Using clean forceps, add 2 DNA Capture Beads to each sample, which should be contained in a Monarch 2 ml Tube.
- Add 550 µl (Low Input: 275 µl) isopropanol, close the cap, and mix on a vertical rotating mixer at 10 rpm for 5 minutes to attach DNA to the beads. If a vertical rotating mixer is not available, invert slowly and gently by hand 25–30 times. A manual inversion is complete when the tube returns to the upright position. Slow inversion is critical for the DNA to bind to the beads; each full inversion should take ~5–6 seconds. If necessary, flick the tube to release any beads that stick to the bottom of the tube.
After a 2–3 inversions, the solution becomes more viscous and the DNA will wrap loosely around the beads. During the following inversions, precipitation of gDNA may be visible, especially with larger input samples. The DNA complex will often contain small air bubbles. With increasing number of inversions, the DNA will completely wrap around the beads, often causing the beads to stick together. DNA binding to the beads should be complete after 25–30 inversions, and the solution should no longer be viscous. Additional inversions may be necessary for larger input samples.
- Remove and discard liquid by pipetting. Avoid removing any of the gDNA wrapped around the glass beads. For optimal DNA solubility, avoid letting the bound DNA dry out on the beads during this and the following steps; add the next buffer quickly. There are two suggested options for carrying out this step:
- Keeping tube upright, insert pipette tip and gently push beads aside to remove liquid.
- Angle the tube so that beads remain at the bottom, and liquid reaches toward tube opening. Pipette from the liquid surface and continue to angle as liquid is removed (tube will be almost horizontal at the end).
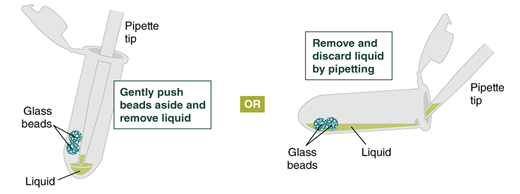
- Keeping tube upright, insert pipette tip and gently push beads aside to remove liquid.
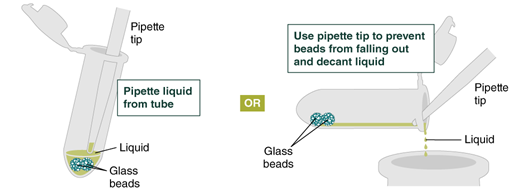
- Add 500 µl gDNA Wash Buffer, close the cap, and mix by inverting the tube 2–3 times. Remove the gDNA Wash Buffer as described in step 3. The loose gDNA complex will condense around the beads more tightly.
- Repeat the wash in Step 4 and remove the gDNA Wash Buffer by pipetting. Alternatively, the buffer can be removed by decanting: position a pipette tip at the top of the angled tube to prevent the beads from falling out. It is not necessary to remove all the gDNA Wash Buffer at this point.
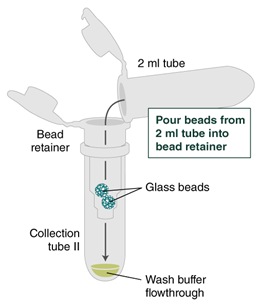
- Place a labeled bead retainer into a Monarch Collection Tube II. Pour the beads into the bead retainer and close the cap. Discard the used Monarch 2 ml Tube. When working with multiple samples, be sure to close the cap of the bead retainer after each transfer of beads.
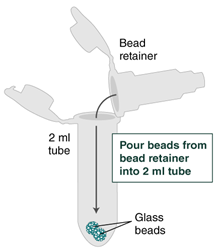
- Pulse spin (≤ 1 second) the sample in a benchtop minicentrifuge to remove any residual wash buffer from the beads.
- Separate the bead retainer from the collection tube, pour the beads into a new, labeled Monarch 2 ml Tube, and insert the used bead retainer into the labeled 1.5 ml microfuge tube (DNA low bind recommended, not provided) for later use during elution. Discard the used collection tube.
- Immediately add 100 µl Elution Buffer II onto the glass beads and incubate for a minimum of 5 minutes at 56°C in a thermal mixer with agitation at the lowest speed (300 rpm). Halfway through the incubation, ensure the beads are not stuck to the bottom of the tube by tilting the tube almost horizontally and gently shaking. This ensures that the beads can move freely, allowing for optimal release of the DNA from the beads. It also ensures that the lower bead does not stick to the bottom of the tube during the following transfer step. Elution volume can be reduced to as low as 50 μl without affecting recovery. However, if using < 100 μl, the gentle shaking of the sample should be done several times during the incubation to ensure complete wetting of the beads.
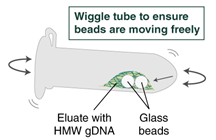
- Ensure the bead retainer is inserted into the 1.5 ml microfuge tube. Pour the eluate and the glass beads into the bead retainer and close the cap. When working with more than 1 sample, it is important to close the cap after each transfer of beads. Typically, all the eluate flows into the bead retainer upon pouring. If any volume remains in the 2 ml tube, spin briefly and transfer.
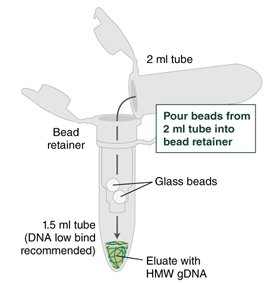
- Centrifuge for 30 seconds at 12,000 x g to separate the eluate from the glass beads. Discard the beads and retainer.
- Pipette eluate up and down 5–10 times with a wide bore pipette tip and ensure any visible DNA aggregates are dispersed. Before analysis or downstream use, HMW DNA must be homogeneously dissolved. After pipetting, incubate at 37°C for 30-60 minutes, overnight at room temperature, or for > 24 hours at 4°C. Pipette up and down 5-10 times again before analyzing or using the HMW DNA. Samples processed using low agitation speeds during lysis will require additional time to fully dissolve. See additional guidance in “Homogenization of HMW DNA Samples”. Samples can be stored at 4°C for short term use (weeks), or at -20°C for long term storage. The elution buffer (10 mM Tris, pH 9.0, 0.5 mM EDTA) is formulated for long term storage of gDNA.