Gibson Assembly® - Building a Synthetic Biology Toolset
In the quest to create the first bacterial cell controlled by a synthetic genome, the J. Craig Venter Institute (JCVI), with support from Synthetic Genomics, Inc. (SGI), developed a variety of powerful new DNA synthesis and assembly methodologies (1–5) to manipulate large, complex DNAs. These methods include a simple, one-step isothermal in vitro recombination technology capable of joining DNAs ranging from relatively short oligonucleotides to fragments hundreds of kilobases in length. This approach, commonly referred to as “Gibson Assembly,” is now being used in laboratories around the world to construct DNA fragments. It has the potential to improve upon traditional cloning methods and opens up a range of innovative and ultimately very useful real-world applications.
Daniel G. Gibson, Ph.D., Synthetic Genomics, Inc. and
Salvatore Russello, Ph.D., New England Biolabs, Inc.
How Gibson Assembly Works
The use of recombinant DNA technology began soon after the discovery of DNA ligase and restriction endonucleases. Soon after, advent of the polymerase chain reaction (PCR) opened up new possibilities for amplification of specific DNA sequences from a complex mixture of genomic DNA. These technologies have been a mainstay in the modern scientific laboratory for several decades and remain useful methods for cloning potentially valuable or interesting DNA today. However, as scientists seek to work with larger DNA fragments, conduct extensive re-engineering of genetic elements, synthesize whole genomes and move towards automated approaches, the technologies required to manipulate DNA also need to evolve.
Investigators at the J. Craig Venter Institute (JCVI) have developed a number of in vitro enzymatic strategies to assemble short oligonucleotides into larger double-stranded DNA constructs (1-4). In 2003, JCVI made a significant advancement in the production of a synthetic genome by assembling the 5,386 bp genome of phiX174, a virus that infects bacteria, in just 14 days (5). This approach involved joining synthetic oligonucleotides by polymerase cycling assembly, and subsequently amplifying them by PCR (5-6). The unprecedented speed with which this was completed laid the foundation for constructing larger and more complex genomes.
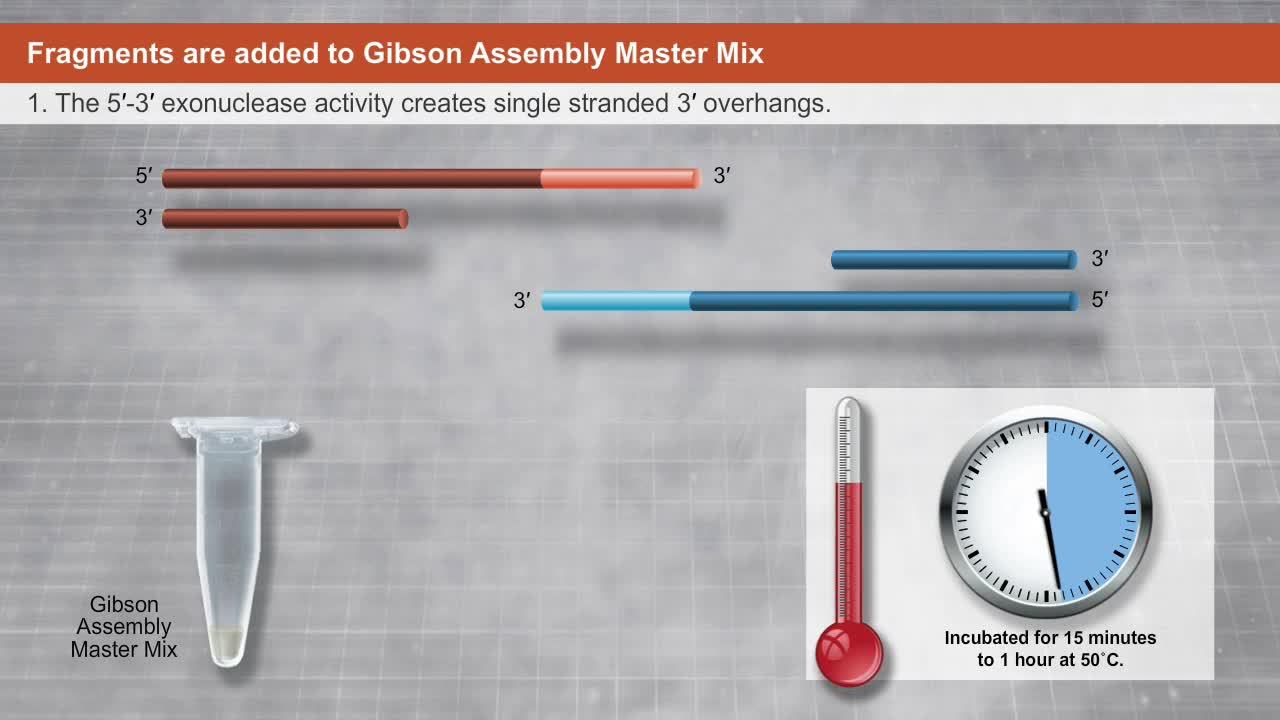
In 2004, JCVI began synthesizing the Mycoplasma genitalium genome. It was found that overlapping DNA molecules could be efficiently joined using three enzyme specificities: (i) exonuclease activity, that chews back the ends of DNA fragments and exposes ssDNA overhangs that can anneal to their ssDNA complement; (ii) DNA polymerase activity, that fills gaps in the annealed products, and (iii) DNA ligase activity, that covalently seals the resulting nicks in the assembly. A two-step thermocycle-based in vitro recombination method utilizing these enzymes was used to join 101 overlapping DNA cassettes into four parts of the M. genitalium genome, each between 136 kb and 166 kb in size. This milestone marked the first assembly of a genome derived from a free-living organism. At 582,970 bp, this synthetic genome was the largest chemically defined DNA structure synthesized in a laboratory, and was 18 times larger than any DNA that had previously been synthesized (4).
Since then, two additional in vitro recombination methods have been developed by JCVI to join and clone DNA molecules larger than 300 kb in a single step (2-4). The simplest of these methods is Gibson Assembly, a one-step isothermal approach that utilizes the same three enzymatic activities described previously. This method can be used to join both ssDNA and dsDNAs.
Figure 1. Overview of Gibson Assembly.
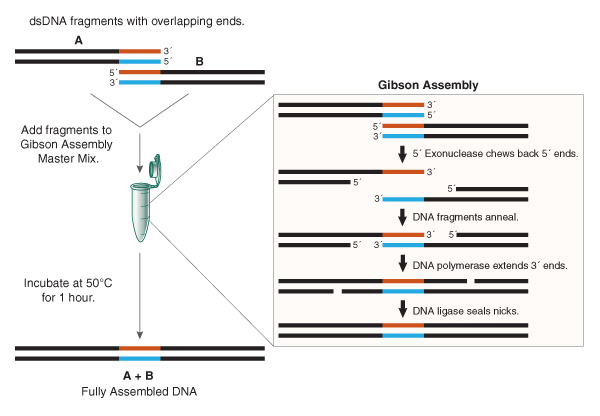
Gibson Assembly has become the most commonly used of the in vitro assembly methods discussed above, as it is easy-to-use, flexible and needs little or no optimization, even for large, complex assemblies. All that is required is input DNA with appropriate overlaps, and an appropriate mix of the three enzymes – the Gibson Assembly Master Mix. DNA fragments are added to the master mix and incubated at 50°C for 1 hour; the resulting assembly product is a fully sealed dsDNA suitable for a range of downstream applications (Figure 1). JCVI has used Gibson Assembly to rapidly synthesize the entire 16,520 bp mouse mitochondrial genome from 600 overlapping 60-base oligonucleotides (3). It was also used in combination with yeast assembly to synthesize the 1.1 Mbp Mycoplasma mycoides genome, which was then activated in a recipient cell to produce the first synthetic cell (1).
Applications of Gibson Assembly
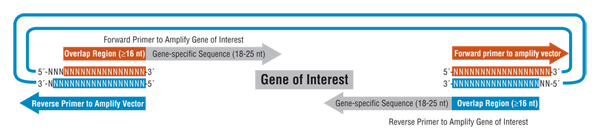
Gibson Assembly eliminates the need to engineer restriction enzyme cut sites within DNA when assembling fragments together. DNA molecules are designed such that neighboring fragments contain a 20-40 bp overlapping sequence. If the DNA fragments originate from PCR products, the overlapping sequence is introduced at the 5′ ends of the primers used in the amplification reaction (Figure 2). DNA fragments can also be assembled with restriction enzyme digested or PCR amplified vector to form circular products suitable for cloning, or for use in downstream applications, such as rolling circle amplification (RCA).
To produce these vectors by PCR, each primer needs to include an overlap with one end of the vector, a restriction site (e.g., Not I) not present within the insert or inserts to enable it to be released from the vector, and an overlap with the ends of the DNA fragment assembly or insert. JCVI has been using this approach to combine DNA fragments with vectors, which are then transformed into E.coli. One or more fragments have been routinely assembled with general cloning vectors, such as pUC19, and assembled into NEB’s pTYB1 expression vector (NEB #N6701). The latter approach was used to express several methylase genes, which aided the genome transplantation efforts at JCVI (8).
Assembly of large DNA constructs.
Laboratories worldwide are beginning to explore the use of synthetic biology approaches in the production of pharmaceuticals, industrial compounds, antibiotics, cosmetics and alternative energy sources (7). This often requires the assembly of a genetic pathway consisting of multiple enzymes and their associated regulatory elements.
Although template DNA is still required, Gibson Assembly simplifies construction of these types of molecules from component fragments. A long stretch of desirable DNA sequence (e.g., a 40 kb genetic pathway) can be broken down into several overlapping PCR products (e.g., eight, 5 kb pieces), which can then be amplified by conventional PCR and combined using Gibson Assembly. This approach has been used to move genetic pathways from one organism to another and to rapidly swap genes, promoters, terminators and ribosome binding sites. DNAs up to ~1 Mbp have been assembled in vitro using Gibson Assembly (2)
Assembly of chemically-synthesized oligonucleotides into dsDNA fragments.
Gibson Assembly can also be used to directly assemble oligonucleotides into a cloning vector, such as pUC19 (3). A common problem observed when chemically synthesizing long stretches of oligonucleotides is the introduction of errors (9). To ensure that error-free molecules are obtained at a reasonable efficiency, a strategy employed by SGI and JCVI involves the assembly of only eight to twelve 60-base oligonucleotides (with 30 bp overlaps) at one time. The resulting dsDNA molecules are sequence-verified and assembled into larger DNA fragments using the same approach.
Because assembly itself does not generally introduce new errors, the final assembled product can be retrieved at high efficiencies. Using this approach, many of the costly and time consuming steps currently used to synthesize DNA, including PCR and an error correction, are eliminated.
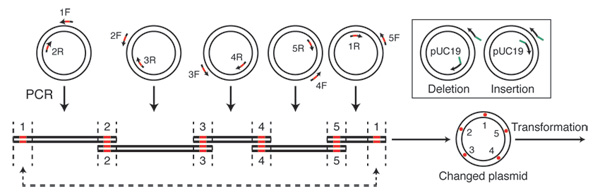
Gibson Assembly can also be used to make rapid changes to DNA fragments, including substitutions, deletions and insertions. To use Gibson Assembly for mutagenesis, the desired changes are introduced into the PCR primers, within the overlapping sequences at assembly points (Figure 3). To modify a DNA sequence in this way, two PCR primers are required: the first contains the desired nucleotide changes, and the second contains the reverse complement of the first primer at the overlapping region. Following amplification and assembly of the fragments, the designed changes are incorporated into the final product. The number of changes that can be made at once depends on the number of fragments simultaneously assembled. For example, an eightpiece assembly, which contains eight assembly points, provides eight opportunities to introduce changes in the DNA sequence. Because the method can be used to assemble large DNA fragments, mutations can rapidly be made to very large pieces of DNA. For example, eight modifications can be introduced into an 80 kb DNA molecule following the assembly of eight 10 kb PCR fragments.
This site-directed mutagenesis strategy was used during synthesis of the M. mycoides genome. The cassettes comprising the synthetic genome were ordered based on an imperfect draft sequence, which resulted in small differences between the synthetic cassettes and the desired M. mycoides genome sequence. The sequences of 16 cassettes were successfully edited using this approach (1).
Figure 2. PCR-Generated Vector and Insert Assembly.
Combinatorial synthesis of DNA Fragments.
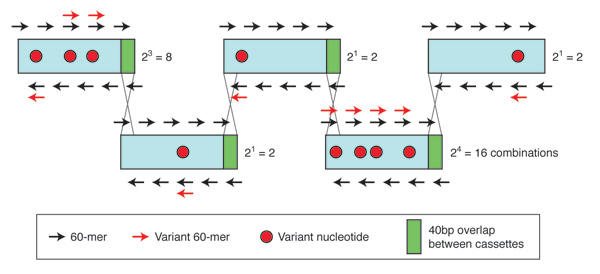
Soon, chromosomes will be designed and synthesized for processes ranging from biofuel production to pharmaceutical manufacture. Bacteria and plants often carry out syntheses that far exceed what can be readily achieved by the best organic chemists. The genes that control desirable pathways can be chemically synthesized, placed in artificial chromosomes, and “installed” in suitable host cells, including bacteria, yeast or plant cells. These multigene pathways can be constructed in a combinatorial fashion, such that each member of the library has a different combination of gene variants. Using screening and selection methods, cells bearing the pathway with the desirable trait (highest yield of a compound, for instance) can be obtained. The engineered host organism then becomes a biologic factory used to manufacture the product specified by the synthetic pathway. Gibson Assembly has the potential to be used to produce combinatorial libraries of synthetic or semisynthetic chromosomes carrying thousands of genes. Figure 4 demonstrates the combinatorial assembly of cassettes produced from 60-mer oligonucleotides. Here, 1,024 (210) variants of a 1 kb gene, containing 10 single nucleotide changes, are produced from 30 sequence-verified cassettes.
Moving Forward
Synthetic & Minimal Cells.
For the past 17 years, the genomes of many organisms have been sequenced and deposited in databases. It has recently been shown that it is possible to reverse this process and synthesize bacterial cells from digitized information (1). In order to realize this vision, researchers at JCVI needed tools and technologies to sequence, synthesize and transplant genomes. Although many hurdles needed to be overcome, synthetic cells can now be produced in the laboratory. As proof of concept, the 1.08 Mbp M. mycoides JCVI-syn1.0 genome was designed, synthesized and assembled, starting from the digitized genome sequence, and transplanted into a Mycoplasma capricolum recipient cell to create new M. mycoides cells controlled only by the synthetic chromosome. The only DNA present in the cells is the designed synthetic DNA, including “watermark” sequences, and other designed gene deletions and polymorphisms and mutations acquired during the building process. The new cells have the expected phenotypic properties and are capable of continuous self-replication (1). The M. mycoides genome is currently the largest chemically defined DNA structure that has been synthesized in a laboratory. It is almost twice as large as the synthetic M. genitalium genome reported in 2008, and more than an order of magnitude larger than any reported DNA sequence synthesized outside JCVI. What has been learned in this “proof of concept” experiment can now be applied to designing and producing new organisms with useful properties.
Further, researchers at JCVI have already begun working on their ultimate objective: to synthesize a minimal cell with only the machinery necessary for independent life. Now that a living cell can be produced from a synthetic genome, components of a synthetic genome can be removed and transplanted in an iterative fashion until only the essential genes are present and the genome is as small as possible. This will help to better understand the function of every gene in a cell and what DNA is required to sustain life in its simplest form. Gibson Assembly is one of the core technologies that will be used to achieve these goals.
Figure 3. Introducing changes to a desired DNA using Gibson Assembly.
Conclusion
Gibson Assembly is a simple and robust method that enables the simultaneous production of many different combinations of genes and pathways, accelerating the progress of synthetic biology. Furthermore, this powerful technology has the potential to help turn DNA sequence into genes and pathways useful in the production of biofuels, industrial compounds, pharmaceuticals and vaccines.
The synthesis of genes and pathways, and even small genomes, has been made easier with Gibson Assembly, helping to move the field of synthetic biology forward. As the power of DNA sequencing increases and sequencing costs decrease, DNA databases will continue to fill with novel genes and pathways waiting to be identified, optimized and expressed in a heterologous host organism. It is time to better understand how to turn these DNA sequences into useful applications.
The ability to quickly construct whole genes and genomes has the potential to accelerate research in a variety of other fields. This capability may also make it possible to quickly respond to emerging threats, and may allow researchers to understand how “life” works. The power of large scale DNA synthesis will dramatically impact the way research is done and vastly accelerate the pace of science. The Gibson Assembly Master Mix provides a new and powerful tool for biotechnology, whose most far-reaching benefits may not yet even be envisioned.
Figure 4. Combinatorial gene synthesis.
Use the NEBuilder Assembly Tool to design primers for your HiFi DNA or Gibson Assembly reactions, based on sequences and polymerase used for amplification.
Additional Information:
- Find more about NEBuilder HiFi DNA Assembly or Gibson Assembly.
- NEBuilder HiFi DNA Assembly Cloning Kit / Master Mix Manual (PDF)
- Gibson Assembly Cloning Kit Manual (PDF)
- Gibson Assembly Master Mix Manual (PDF)
- Gibson, D.G. et al. (2010) Science, 239, 52–56.
- Gibson, D.G. et al. (2009) Nature Methods, 6, 343–345.
- Gibson, D.G. et al. (2010) Nature Methods, 7, 901–903.
- Gibson, D.G. et al. (2008) Science, 319, 1215–1220.
- Smith et al. (2003) PNAS, 100, 15440–15445.
- Stemmer, W.P. et al. (1995) Gene, 164, 49–53.
- Endy, D. (2005) Nature, 438, 449–453.
- Lartique, C. et al. (2009) Science, 325, 1693–1696.
- Carr et al. (2004) Nucleic Acids Res. 32, e162.

