Protocol for use with NEBNext Direct Genotyping Solution (NEB #E9500 and #E9530)
Symbols
| This is a point where you can safely stop the protocol and store the samples prior to proceeding to the next step in the protocol. | |
| This caution sign signifies a step in the protocol that has two paths leading to the same end point. | |
| Colored bullets indicate the cap color of the reagent to be added. |
NEBNext Direct Genotyping Solution Sample Prep Kit (NEB #E9500B-S)
1.1. Quantify DNA
1.1.1. Quantify input DNA using a Qubit dsDNA HS Assay Kit, Quant-iT PicoGreen dsDNA Assay Kit, Agilent TapeStation, or similar assay. Do not use a UV-based assay for quantification.
1.2. Fragmentation and End Prep
Note: For step 1.2 (Fragmentation and End Prep), we recommend making a master mix using 20% more volume than required per reaction. For steps 1.6 (Bait Hybridization), 1.9 (3’ Blunting of DNA), 1.10 (3’ Adaptor Ligation), and 1.12 (Library Amplification), we recommend making master mixes using 10% more volume than required per reaction. Overage is included for each component to accommodate excess for master mixes.
1.2.1. Vortex the Fragmentation and End Prep Enzyme Mix thoroughly for 5-8 seconds prior to use.
Note: It is important to vortex the enzyme mix prior to use for optimal performance.
1.2.2. Set up the following reaction on ice, in either a 384-well plate or a 96-well plate. DNA should be diluted in pre-chilled, sterile, nuclease-free water to a total volume of 6 µl. Aliquot DNA and water first, then add 2 µl of master-mixed buffer and enzyme to samples.
|
REAGENT |
PER REACTION |
WITH 20% OVERAGE |
|---|---|---|
|
DNA (10-100 ng)* |
6 μl |
N/A |
|
|
1.5 μl |
1.8 μl |
|
|
0.5 μl |
0.6 μl |
|
Total Volume |
8 μl |
N/A |
*See Table 1.2.1 for guidelines on DNA input amounts.
Table 1.2.1: Recommended DNA Input Amounts Based on Haploid Genome Size and Panel Size.
|
HAPLOID GENOME SIZE |
RECOMMENDED DNA INPUT AMOUNT FOR 100-500 SNP PANELS |
RECOMMENDED DNA INPUT AMOUNT FOR 501-5,000 SNP PANELS |
|
< 1 Gb |
10-25 ng |
25-50 ng |
|
> 1 Gb |
25-50 ng |
50-100 ng |
1.2.3. Seal the plate, briefly vortex the samples to mix, and quickly spin down the plate prior to incubation. Alternatively, samples may be mixed by pipetting up and down at least 10 times, then covered with a plate seal and quickly spun down for incubation.
1.2.4. Run the following program in a thermocycler with the heated lid set to 75°C:
15 mins @ 37°C
30 mins @ 65°C
Hold @ 4°C
![]() If necessary, samples can be stored at -20°C for up to 16 hours; however, we recommend continuing with 5′ Adaptor Ligation before stopping.
If necessary, samples can be stored at -20°C for up to 16 hours; however, we recommend continuing with 5′ Adaptor Ligation before stopping.
1.3. 5′ Adaptor Ligation with Sample Indexing
1.3.1. Determine the number of samples (between 4 to 96) that will be pooled together for a single hybridization.
1.3.2. Ensure that you choose a valid combination of barcoded adaptors following guidelines in Section 3, Table 3.1.
1.3.3. Refer to Section 2 for guidelines on using the single-use ![]() (red) NEBNext Direct GS Indexed 5′ Adaptor Plate.
(red) NEBNext Direct GS Indexed 5′ Adaptor Plate.
1.3.4. Briefly spin down the sample plate from Step 1.2.4 to collect samples to the bottom of the wells.
1.3.5. Remove the plate seal and to each sample well add 5 μl of indexed 5′ adaptor from the ![]() (red) NEBNext Direct GS Indexed 5′ Adaptor Plate. The 5′ Adaptor incorporates a unique sample index into each sample prior to pooling. Use one unique indexed adaptor per sample that will be subsequently pooled together.
(red) NEBNext Direct GS Indexed 5′ Adaptor Plate. The 5′ Adaptor incorporates a unique sample index into each sample prior to pooling. Use one unique indexed adaptor per sample that will be subsequently pooled together.
1.3.6. Add 10 μl of ![]() (red) 5′ Adaptor Ligation Master Mix to each sample.
(red) 5′ Adaptor Ligation Master Mix to each sample.
1.3.7. Mix the samples by sealing the plate and vortexing thoroughly, followed by a quick spin, or by pipetting up and down at least 10 times.
1.3.8. Incubate the sample in a thermocycler for 15 minutes at 20°C with the lid unheated.
1.3.9. Remove the plate from the thermocycler and stop the ligation by adding 2.5 μl of stop solution. Mix well by sealing the plate and vortexing thoroughly, followed by a quick spin, or by pipetting up and down at least 10 times.
1.4. Sample Pooling and Bead Cleanup
1.4.1. Combine 24 μl from each uniquely indexed sample into an appropriately sized fresh, sterile, nuclease-free tube(s) based on the total volume specified in Step 1.4.3.
Note: Each pool of samples should contain all uniquely indexed samples intended for a single hybridization reaction.
1.4.2. Immediately before use, vortex the ![]() (brown) Sample Purification Beads thoroughly to resuspend.
(brown) Sample Purification Beads thoroughly to resuspend.
1.4.3. Add ![]() (brown) Sample Purification Beads to each pool of samples for a 0.8X bead cleanup. See below for example volumes of
(brown) Sample Purification Beads to each pool of samples for a 0.8X bead cleanup. See below for example volumes of
Sample Purification Beads to add based on the number of samples pooled:
|
# OF POOLED SAMPLES |
VOLUME OF POOLED SAMPLES |
VOLUME OF SAMPLE PURIFICATION BEADS |
TOTAL VOLUME |
|---|---|---|---|
|
24 |
576 μl |
461 μl |
1,037 μl |
|
47 |
1,128 μl |
902 μl |
2,030 μl |
|
96 |
2,304 μl |
1,843 μl |
4,147 μl |
1.4.4. Mix well by vortexing thoroughly or by pipetting up and down at least 10 times.
1.4.5. Incubate the samples for 5 minutes at room temperature.
![]()
1.4.6. For sample pools ≤ 46 samples: Place the samples on a magnet and wait for the solution to clear.
For sample pools greater > 46 samples: Either a 5 ml or 15 ml tube with the pooled samples and beads can be placed on an appropriate magnet, or the pooled samples and beads can be distributed into multiple 1.5 ml or 2 ml tubes and placed on a tube magnet.
1.4.7. After the solution is clear (about 2 minutes), carefully remove and discard the supernatant. Be careful not to disturb the beads that contain the DNA fragments (Caution: do not discard the beads).
1.4.8. While the tubes are on the magnet, add enough 80% EtOH (freshly prepared) to cover the bead pellet. For example, if the samples are in a 1.5 ml tube, ~1 ml of 80% EtOH is sufficient to cover the bead pellet; if the samples are in a 15 ml falcon tube, ~10 ml of EtOH is required to cover the bead pellet depending on the model of the magnet used.
1.4.9. Incubate the samples at room temperature for 30 seconds, then carefully remove and discard the 80% EtOH. Be careful not to disturb the beads that contain the DNA fragments.
1.4.10. Repeat steps 1.4.8–1.4.9 once for a total of 2 washes in 80% EtOH. Ensure that all visible liquid is removed after the second wash.
1.4.11. Incubate the samples on the magnet at room temperature with the tube caps open for up to 5 minutes to dry the beads.
Caution: Do not over-dry the beads. This may result in lower recovery of DNA. Elute the samples when the beads are still dark brown and glossy looking, but when all visible liquid has evaporated. When the beads turn lighter brown and start to crack they are too dry.
![]()
1.4.12. For sample pools ≤ 46 samples: Remove the tubes from the magnet and resuspend the beads in 200 μl of sterile, nuclease-free water to elute the samples from the beads.
For sample pools > 46 samples: Remove the tubes from the magnet. If the samples and beads were distributed across multiple 1.5 ml or 2 ml tubes for ethanol washes, divide 200 μl of sterile, nuclease-free water across the number of tubes used to resuspend each bead pellet and elute the samples from the beads. If the sample pool and beads were kept in a 5 ml or 15 ml tube, resuspend the beads in 200 μl of sterile, nuclease-free water to elute the samples from the beads.
1.4.13. Incubate for 2 minutes at room temperature.
1.4.14. Place the samples on a magnet and wait for the solution to clear.
![]()
1.4.15. For sample pools ≤ 46 samples: Transfer 198 μl of the eluted sample to a fresh tube.
For sample pools > 46 samples: If the samples and beads were distributed across multiple 1.5 ml or 2 ml tubes for ethanol washes, combine the eluates from the multiple tubes to transfer a combined total of 198 μl of elution into a fresh tube.
1.4.16. Proceed immediately with Step 1.5 using the NEBNext Direct Genotyping Solution Target Enrichment Kit (NEB #E9530), or store samples at -20°C for up to 2 weeks.
![]()
Purified DNA sample pools can be stored for up to 2 weeks at -20°C.
NEBNext Direct Genotyping Solution Target Enrichment Kit (NEB #E9530B-S)
1.5. Pre-hybridization Bead Cleanup
1.5.1 Immediately before use, vortex the ![]() (brown) Pre-Hyb Sample Purification Beads thoroughly to resuspend.
(brown) Pre-Hyb Sample Purification Beads thoroughly to resuspend.
1.5.2 Add 360 μl of ![]() (brown) Pre-Hyb Sample Purification Beads to the pooled samples from Step 1.4.15.
(brown) Pre-Hyb Sample Purification Beads to the pooled samples from Step 1.4.15.
1.5.3. Mix well by vortexing thoroughly or by pipetting up and down 10 times with the pipette set to 500 μl.
1.5.4. Incubate the samples for 5 minutes at room temperature.
1.5.5. Place the samples on a magnet and wait for the solution to clear.
1.5.6. After the solution is clear (about 2 minutes), carefully remove and discard the supernatant. Be careful not to disturb the beads that contain the DNA fragments (Caution: do not discard the beads).
1.5.7. While the tubes are on the magnet, add 1 ml of freshly prepared (same day) 80% EtOH to each tube.
1.5.8. Incubate the samples at room temperature for 30 seconds, then carefully remove and discard the 80% EtOH. Be careful not to disturb the beads that contain the DNA fragments.
1.5.9. Repeat Steps 1.5.7.–1.5.8. once for a total of 2 washes in 80% EtOH. Ensure that all visible liquid is removed after the second wash.
1.5.10. Incubate the samples on the magnet at room temperature with the tube caps open for up to 5 minutes to dry the beads.
Caution: Do not over-dry the beads. This may result in lower recovery of DNA. Elute the samples when the beads are still dark brown and glossy looking, but when all visible liquid has evaporated. When the beads turn lighter brown and start to crack they are too dry.
1.5.11. Resuspend the dry beads in 62 μl of sterile, nuclease-free water to elute the samples from the beads.
1.5.12. Incubate for 2 minutes at room temperature.
1.5.13. Place the samples on a magnet and wait for the solution to clear.
1.5.14. Transfer 60 μl of the eluted sample into a fresh PCR strip tube or plate. Take care not to transfer Sample Purification Beads with the eluted sample.
Note: Any Sample Purification Beads carried over into the hybridization will increase the off-target molecules that are converted into the final library.
1.6. Bait Hybridization
1.6.1. Make a Hybridization master mix by combining the following components for the appropriate number of reactions. Vortex the Hybridization Buffer to mix well prior to pipetting and pipette the buffer slowly to ensure accurate volume delivery.
|
REAGENT |
PER REACTION |
WITH 10% OVERAGE |
|---|---|---|
|
|
40 μl |
44 μl |
|
|
15 μl |
16.5 μl |
|
|
5 μl |
5.5 μl |
|
Total Volume |
60 μl |
66 μl |
1.6.2. Mix the master mix well by vortexing for 3–5 seconds and centrifuge briefly.
1.6.3. To each pooled sample from Step 1.5.14, add 60 μl of Hybridization master mix for a final volume of 120 μl.
1.6.4. Seal the PCR plate or cap tubes securely to avoid evaporation. Mix well by briefly vortexing, followed by a quick spin. Alternatively, samples may be mixed by pipetting up and down 10 times prior to capping the tubes/sealing the plate for incubation.
1.6.5. Run the following program with the heated lid set to 105°C and place the samples in the thermocycler after the block temperature reaches 95°C. Proceed to Section 1.7 during this incubation.
10 mins @ 95°C
90 mins @ 60°C
Hold @ 60°C
1.6.6. While the samples are incubating, prepare ![]() (blue) Streptavidin beads (see Streptavidin Bead Preparation in Section 1.7).
(blue) Streptavidin beads (see Streptavidin Bead Preparation in Section 1.7).
1.6.7. After the 90 min incubation at 60°C and when Section 1.7 (Streptavidin Bead Preparation) is complete, unseal the tubes/wells, leave the samples on the thermocycler at 60°C with the lid open and proceed to Bead Binding in Section 1.8.
1.7. Streptavidin Bead Preparation
1.7.1. Equilibrate the ![]() (blue) Streptavidin Beads to room temperature (~15 minutes).
(blue) Streptavidin Beads to room temperature (~15 minutes).
1.7.2. Immediately before use, vortex the ![]() (blue) Streptavidin Beads thoroughly to resuspend.
(blue) Streptavidin Beads thoroughly to resuspend.
1.7.3. Multiply the number of hybridizations performed in Step 1.6 by 82.5 μl, and transfer that volume of ![]() (blue) Streptavidin Beads into a sterile, nuclease-free tube. See table (below) for example volumes.
(blue) Streptavidin Beads into a sterile, nuclease-free tube. See table (below) for example volumes.
|
# OF HYBRIDIZATION REACTIONS |
VOLUME OF BEADS |
VOLUME OF HYBRIDIZATION WASH (HW) |
VOLUME OF BEAD PREP BUFFER |
|---|---|---|---|
|
2 |
165 μl |
330 μl |
66 μl |
|
4 |
330 μl |
660 μl |
132 μl |
|
8 |
660 μl |
1320 μl |
264 μl |
1.7.4. Place the tube on a magnet and wait for the solution to clear (~1 minute). Remove the supernatant, and then remove the tube from the magnet.
1.7.5. Multiply the number of hybridizations performed in Step 1.6 by 165 μl and transfer that volume of ![]() (blue) Hybridization Wash (HW) to the beads. See table in Step 1.7.3 for example volumes. Resuspend the beads by vortexing thoroughly followed by a quick spin or by pipetting up and down 10 times.
(blue) Hybridization Wash (HW) to the beads. See table in Step 1.7.3 for example volumes. Resuspend the beads by vortexing thoroughly followed by a quick spin or by pipetting up and down 10 times.
1.7.6. Place the tube on a magnet and wait for the solution to clear (~1 minute). Remove the supernatant, and then remove the tube from the magnet.
1.7.7. Repeat Steps 1.7.5–1.7.6 once for a total of 2 washes with ![]() (blue) Hybridization Wash (HW).
(blue) Hybridization Wash (HW).
1.7.8. Multiply the number of hybridizations performed in Step 1.6 by 33 μl and transfer that volume of ![]() (blue) Bead Prep Buffer to the beads. See table in Step 1.7.3 for example volumes. Vortex the beads in Bead Prep Buffer to resuspend.
(blue) Bead Prep Buffer to the beads. See table in Step 1.7.3 for example volumes. Vortex the beads in Bead Prep Buffer to resuspend.
1.7.9. Keep the beads in Bead Prep Buffer at room temperature until Bait Hybridization (Section 1.6) is completed.
Note: For Sections 1.8–1.11 (Bead Binding through Off-Target Removal), the thermocycler lid should be unheated. The lid can remain open and PCR tubes can remain uncapped for convenience. However, if it is preferred, tubes can be capped during incubations and the lid can be closed once the lid temperature has cooled down from hybridization.
1.8 Bead Binding
1.8.1. Immediately before use, vortex the washed Streptavidin Beads (from Step 1.7.9) in Bead Prep Buffer to resuspend.
1.8.2. Add 30 μl of resuspended beads to each hybridization reaction (from Step 1.6.7) while the samples are on the thermocycler at 60°C and mix thoroughly by pipetting up and down 10 times.
Note: If you have a large number of hybridizations, the Streptavidin Beads may start to settle as you distribute the beads to each reaction. To prevent this, pipette up and down or vortex the beads frequently to keep them in suspension while pipetting into individual reactions.
1.8.3. Change the thermocycler temperature to 48°C with the heated lid off and incubate the reactions for 10 minutes.
1.8.4. Remove the samples from the thermocycler and place on a magnet. Wait for the solution to clear (~15 seconds), remove the supernatant, and then remove the samples from the magnet.
1.8.5. Add 150 μl of ![]() (blue) Hybridization Wash (HW) to each sample. Mix by pipetting up and down 10 times. Place the samples on a thermocycler set at 62°C and incubate for 5 minutes.
(blue) Hybridization Wash (HW) to each sample. Mix by pipetting up and down 10 times. Place the samples on a thermocycler set at 62°C and incubate for 5 minutes.
1.8.6. Repeat Steps 1.8.4–1.8.5 once for a total of 2 washes at 62°C.
1.8.7. Remove the samples from the thermocycler and place on a magnet. Wait for the solution to clear (~15 seconds), remove the supernatant, and then remove the samples from the magnet.
1.8.8. Add 150 μl of ![]() (light pink) Bead Wash Buffer 2 (BW2) to each sample. Mix by pipetting up and down 10 times.
(light pink) Bead Wash Buffer 2 (BW2) to each sample. Mix by pipetting up and down 10 times.
![]()
Samples can be kept at room temperature in BW2 buffer for up to 30 minutes. If longer storage is required, place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet. Resuspend the beads in 100 μl of 1X TE (provided). DNA-bound beads can be stored in 1X TE for up to 24 hours at 4°C (Caution: do not freeze). When ready to proceed with the protocol, place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet. Add 150 μl of BW2 to each reaction, mix gently by pipetting up and down 10 times, and then proceed directly to 3′ Blunting of DNA in Section 1.9.
1.9. 3′ Blunting of DNA
1.9.1. While the beads are suspended in BW2 buffer, make a 3′ Blunting master mix by adding the following components in a sterile, nuclease-free tube for the appropriate number of reactions. Vortex the 3′ Blunting Buffer thoroughly to mix well prior to pipetting, and pipette the buffer slowly to ensure accurate volume delivery.
|
REAGENT |
PER REACTION |
WITH 10% OVERAGE |
|---|---|---|
|
|
97 μl |
106.7 μl |
|
|
3 μl |
3.3 μl |
|
Total Volume |
100 μl |
110 μl |
1.9.2. Mix the master mix well by vortexing thoroughly for 3–5 seconds and centrifuge briefly.
1.9.3. Place the DNA-bound beads on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet.
1.9.4. Add 100 μl of 3′ Blunting master mix (from Step 1.9.1) to each sample. Mix thoroughly by pipetting up and down 10 times. Incubate the samples at 37°C for 10 minutes. Ensure that the thermocycler lid has cooled to below 37°C, or keep the thermocycler lid open.
Note: For optimal performance, it is important that the incubation time for this step is closely followed.
1.9.5. Proceed immediately with the Post-reaction Wash (Section 1.9.6).
1.9.6.1. Place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet.
1.9.6.2. Add 150 μl of ![]() (dark purple) Bead Wash Buffer 1 (BW1) to each sample. Mix by pipetting up and down 10 times. Place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet.
(dark purple) Bead Wash Buffer 1 (BW1) to each sample. Mix by pipetting up and down 10 times. Place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet.
1.9.6.3. Add 150 μl of ![]() (light pink) Bead Wash Buffer 2 (BW2) to each sample. Mix by pipetting up and down 10 times.
(light pink) Bead Wash Buffer 2 (BW2) to each sample. Mix by pipetting up and down 10 times.
![]()
Samples can be kept at room temperature in BW2 buffer for up to 30 minutes. If longer storage is required, place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet. Resuspend the beads in 100 μl of 1X TE (provided). DNA-bound beads can be stored in 1X TE for up to 24 hours at 4°C (Caution: do not freeze). When ready to proceed with the protocol, place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet. Add 150 μl of BW2 to each reaction, mix gently by pipetting up and down 10 times, and then proceed directly to 3′ Adaptor Ligation in Section 1.10.
1.10. 3′ Adaptor Ligation
1.10.1. While the beads are suspended in BW2 buffer, make a 3′ Adaptor Ligation master mix by adding the following components in a sterile, nuclease-free tube for the appropriate number of reactions. Vortex the Adaptor Ligation Buffer thoroughly to mix well prior to pipetting and pipette the buffer slowly to ensure accurate volume delivery.
|
REAGENT |
PER REACTION |
WITH 10% OVERAGE |
|---|---|---|
|
|
80 μl |
88 μl |
|
|
10 μl |
11 μl |
|
|
10 μl |
11 μl |
|
Total Volume |
100 μl |
110 μl |
1.10.2. Mix the master mix by vortexing thoroughly for 3–5 seconds and centrifuge briefly.
1.10.3. Place the DNA-bound beads on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet.
1.10.4. Add 100 μl of 3′ Adaptor Ligation master mix (from Step 1.10.1) to each sample. Mix thoroughly by pipetting up and down 10 times.
1.10.5. Incubate the samples at 20°C for 15 minutes on a thermocycler with the thermocycler lid unheated.
1.10.6. When the 15 minute incubation is complete, proceed immediately to Off-Target Removal (Step 1.11). Do not wash the beads in between 3′ Adaptor Ligation and Off-Target Removal.
1.11. Off-Target Removal
1.11.1. To each sample from Step 1.10.6, add 5 μl of ![]() (lilac) Specificity Enhancer Enzyme Mix.
(lilac) Specificity Enhancer Enzyme Mix.
1.11.2. Mix thoroughly by pipetting up and down at least 10 times.
1.11.3. Change the thermocycler temperature to 37°C with the thermocycler lid unheated and incubate the samples for 15 minutes.
1.11.4. Proceed immediately with the Post-reaction Wash (Section 1.11.5).
1.11.5. Post-reaction Wash
1.11.5.1. Place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet.
1.11.5.2. Add 150 μl of ![]() (dark purple) Bead Wash Buffer 1 (BW1) to each sample. Mix by pipetting up and down 10 times. Place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet.
(dark purple) Bead Wash Buffer 1 (BW1) to each sample. Mix by pipetting up and down 10 times. Place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet.
1.11.5.3. Add 150 μl of ![]() (light pink) Bead Wash Buffer 2 (BW2) to each sample. Mix by pipetting up and down 10 times.
(light pink) Bead Wash Buffer 2 (BW2) to each sample. Mix by pipetting up and down 10 times.
![]()
Samples can be kept at room temperature in BW2 buffer for up to 30 minutes. If longer storage is required, place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet. Resuspend the beads in 100 μl of 1X TE (provided). DNA-bound beads can be stored in 1X TE for up to 24 hours at 4°C (Caution: do not freeze). When ready to proceed with the protocol, place the samples on a magnet and wait for the solution to clear (~15 seconds). Remove the supernatant, and then remove the samples from the magnet. Add 150 μl of BW2 to each reaction, mix gently by pipetting up and down 10 times, and then proceed directly to Library Amplification in Section 1.12.
1.12. Library Amplification
Refer to Section 3, Table 3.2 for guidelines on index pooling.
1.12.1. For each hybridization, make a Library Amplification master mix by combining the following components in a sterile, nuclease- free tube for the appropriate number of reactions. Mix the Q5 Master mix well prior to pipetting.
|
REAGENT |
PER REACTION |
WITH 10% OVERAGE |
|---|---|---|
|
|
50 μl |
55 μl |
|
Sterile, nuclease-free water |
45 μl |
49.5 μl |
|
Total Volume |
95 μl |
104.5 μl |
1.12.2. Place the samples on a magnet, wait for the solution to clear (~15 seconds), remove the supernatant then remove the samples from the magnet.
1.12.3 Move the samples to ice and add 95 μl of the Library Amplification master mix and 5 μl of (dark blue) indexed primer mix for a total volume of 100 μl for the PCR reaction. The indexed primer mix incorporates the pool index into each pool of samples that were processed through the NEBNext Direct GS Target Enrichment Kit as a single tube. Use one unique indexed primer mix per reaction.
1.12.4 Mix thoroughly by pipetting up and down 10 times, then cap the tubes or seal the plate.
1.12.5. Run the following program with the heated lid set to 105°C and place the samples in the thermocycler when the block temperature reaches 98°C:
|
CYCLE STEP |
TEMP |
TIME |
CYCLES |
|---|---|---|---|
|
Initial Denaturation |
98°C |
30 seconds |
1 |
|
Denaturation |
98°C |
10 seconds |
12-21* |
|
Annealing/Extension |
65°C |
75 seconds |
|
|
Final Extension |
65°C |
5 minutes |
1 |
|
Hold |
4°C |
∞ |
|
*Follow the PCR cycle number recommendations listed in Table 1.12.1 and Table 1.12.2.
The following guidelines should be used to determine the number of PCR cycles. To determine the total input in a hybridization, multiply the input amount per sample by the # of pooled samples per hybridization. For example, the total input in a hybridization of 96 pooled samples with 25 ng of input each is 2,400 ng and would require 15 PCR cycles for a 400 Mb genome and a 500 SNP panel.
The recommended cycle numbers listed should be used as a starting point to produce sufficient library concentrations for a sequencing run while avoiding PCR artifacts and over-cycling (high molecular-weight fragments on a Bioanalyzer or TapeStation). Some optimization may be required.
Table 1.12.1: PCR Cycle Number Recommendations for 100-1,000 SNP Panels
|
TOTAL INPUT IN THE HYBRIDIZATION |
HAPLOID GENOME SIZE |
||
|---|---|---|---|
|
400 Mb |
3 Gb |
15 Gb |
|
|
240-800 ng |
17 |
19 |
21 |
|
800-1,200 ng |
16 |
18 |
20 |
|
1,200-2,400 ng |
15 |
17 |
19 |
|
2,400-4,800 ng |
14 |
16 |
18 |
|
4,800-9,600 ng |
13 |
15 |
17 |
Table 1.12.2: PCR Cycle Number Recommendations for 1,001-5,000 SNP Panels
|
TOTAL INPUT IN THE HYBRIDIZATION |
HAPLOID GENOME SIZE |
||
|---|---|---|---|
|
400 Mb |
3 Gb |
15 Gb |
|
|
240-800 ng |
16 |
18 |
20 |
|
800-1,200 ng |
15 |
17 |
19 |
|
1,200-2,400 ng |
14 |
16 |
18 |
|
2,400-4,800 ng |
13 |
15 |
17 |
|
4,800-9,600 ng |
12 |
14 |
16 |
1.12.6. Proceed to Purify and Size Select Amplified Fragments in Section 1.13.
PCR products with beads can be stored for up to 24 hours at 4°C (Caution: do not freeze).
1.13. Purify and Size Select Amplified Fragments
1.13.1. If you detect significant evaporation in any of the PCR reactions, bring the volume up to 100 μl with sterile, nuclease-free water.
1.13.2. Immediately before use, vortex the ![]() (brown) Post-PCR Sample Purification Beads to resuspend.
(brown) Post-PCR Sample Purification Beads to resuspend.
1.13.3. Add 85 μl of Post-PCR Sample Purification Beads to each PCR reaction. Mix well by pipetting up and down at least 10 times.
1.13.4. Incubate for 5 minutes uncapped at room temperature.
1.13.5. Place the tubes/PCR plate on a magnet. After the solution is clear (about 2 minutes), carefully remove and discard the supernatant. Be careful not to disturb the beads that contain the DNA targets (Caution: do not discard beads).
1.13.6. While the samples are on the magnet, add 200 μl of freshly prepared (same day) 80% EtOH to each tube.
1.13.7 Incubate at room temperature for 30 seconds, and then carefully remove and discard the 80% EtOH. Be careful not to disturb the beads that contain the DNA fragments.
1.13.8. Repeat steps 1.13.6-1.13.7 once for a total of 2 washes in 80% EtOH. Ensure that all visible liquid is removed after the second wash.
1.13.9. Incubate the samples on the magnet at room temperature uncapped or unsealed for up to 5 minutes to dry the beads.
Caution: Do not over-dry the beads. This may result in lower recovery of DNA. Elute the samples when the beads are still dark brown and glossy looking, but when all visible liquid has evaporated. When the beads turn lighter brown and start to crack they are too dry.1.13.10. Remove the tubes/plate from the magnet and resuspend the dry beads in 30 μl of sterile, nuclease-free water or 1X TE by gently pipetting (or gently vortex capped tubes/sealed plate, followed by a quick spin).
1.13.11. Incubate for 2 minutes at room temperature.
1.13.12. Place the tubes on a magnet and allow the solution to clear.
1.13.13. Transfer 28 μl of the eluted library to a fresh tube.
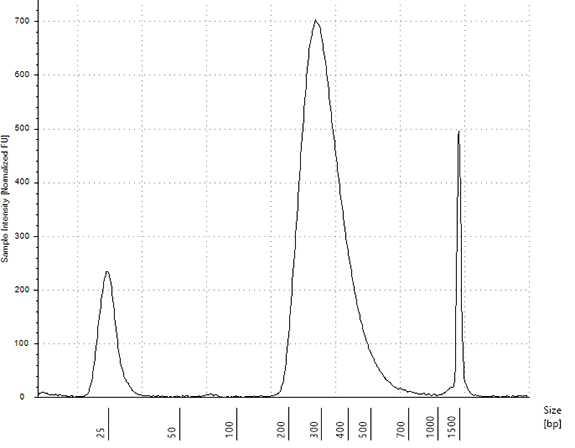
1.13.14. Evaluate 1 μl of the eluted library on an Agilent High Sensitivity D1000 ScreenTape or with a similar assay.