NEB® TV Ep. 1 – Synthetic Biology
Script
Deana Martin, PhD:
Welcome to the first episode of NEB TV. Today, I'm joined by Peter Weigle, one of our staff scientists. Welcome, Peter.
Peter Weigle,PhD:
Hi, Deana. Thank you.
Deana Martin, PhD:
And we are talking about synthetic biology.
Peter Weigle,PhD:
So for today's episode, I'm gonna give you a one minute overview of what is synthetic biology, then we're gonna hear from my colleague, Breton, about the highlights of the SEED synthetic biology meeting, which just happened here locally in Boston. And then we're gonna round up the episode with some thoughts about where synthetic biology is going and we're going to be hearing from Deepak Mishra and Chris Voigt who are both working at MIT.
To me, synthetic biology is programming with DNA, and by programming I mean using recombinant nucleic acids to direct biological systems to execute a subset of predetermined outputs based on the computation of multiple inputs. Program behaviors could be simple, such as the expression of a fluorescent reporter gene in response to some environmental toxin or more complex with sequential stages occurring in temporal sequence, such as an engineered stem cell programmed to adopt one of several predefined cell states based on its position within a tissue into which it's been injected.
The engineering mindset for programming biology in this way borrows heavily from analog circuitry, digital logic, but these are really a framework, a shorthand to facilitate the design of increasingly complex systems of gene regulation. Whatever the approach, the material foundation of synthetic biology is the availability of inexpensive pieces of custom chemically synthesized DNA as well as the enzymatic methods for putting those DNAs together.
Deana Martin,PhD:
So I'm here with Breton Hornblower, one of our product marketing managers, hey Breton.
Breton Hornblower,PhD :
Hey, how's it going Deana?
Deana Martin, PhD:
Good. Breton recently attended the SEED Meeting which NEB sponsored. Breton, can you tell us what SEED stands for?
Breton Hornblower, PhD:
Sure. It stands for Synthetic Biology, Engineering, Evolution and Design.
Deana Martin,PhD:
And what was the most interesting talk that you attended?
Breton Hornblower,PhD:
I think probably the most interesting talk was on a Ron Weiss's talk from MIT. So he described encoding biological circuits in RNA molecules rather than DNA. The primary advantages being that you wouldn't have to integrate with the host genome, that you could also achieve some sort of temporal control for the RNA.
Deana Martin,PhD:
Great. Will you be attending SEED next year?
Breton Hornblower PhD:
We will. It's gonna be in Chicago in June.
Deana Martin,PhD:
Excellent, thank you.
Breton Hornblower PhD:
Thanks.
Deepak Mishra:
So I'm Deepak Mishra.
Christopher Voight, PhD:
My name is Christopher Voigt.
Deepak Mishra:
I'm currently a graduate student in biological engineering at MIT.
Christopher Voight, PhD.:
I'm a professor of biological engineering at the Massachusetts Institute of Technology.
So synthetic biology is about the process of genetic engineering.
Deepak Mishra:
So it's a catch all term of just these are novel functions that a cell may do to some extent, but they may actually be things that we've just completely put in all together and we have engineering control over.
Christopher Voight, PhD.:
And the ambition of the field is to be able to access the natural complexity that we see in the world and not be limited in the types of products that we can use biology to produce.
Deepak Mishra:
If we want to build a molecule or if we want to build a new plastic, we'll get an organism that actually grows and produces that molecule, produces that plastic for us to use.
Christopher Voight, PhD.:
Well what we're seeing is an incredible increase in the scale of genetic engineering projects. So a few years ago, it was really only possible to modify a couple of genes as part of a project. But now, we're looking at major projects that involve dozens of genes with different cellular systems.
Deepak Mishra:
If we have to get a new drug treatment because we're sick, we're gonna go in and a doctor's gonna take some cells from us and actually grow a new little artificial liver that's as big as like a fist and test the various drugs on you to make sure it's actually going to be effective and safe. And we're gonna go all the way where maybe one day you do need to get a skin transplant or an organ transplant and we're gonna encode genetic networks into your very own cells that actually augment your body and are able to do just that.
Christopher Voight,PhD.:
So it's trying to be able to build systems in biology that are larger and more sophisticated than what's been done previously.
Deana Martin, PhD:
So thanks for joining us today, Peter.
Peter Weigle,PhD:
My pleasure.
Deana Martin,PhD:
And if you have any suggestions for future episodes, please let us know.
Related Videos
-

Science in 60 CRISPR/Cas9 -

NEB® TV Ep. 7 – Synthetic Biology/ CRISPR -
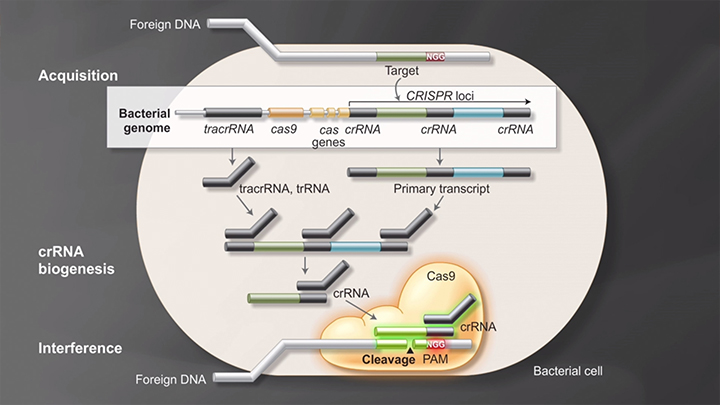
Bacterial Adaptive Immunity with CRISPR/Cas9

