The Next Generation of Reagents for Sample Preparation
Sample preparation is a critical step in a wide variety of analytical techniques in which a sample is biochemically or enzymatically treated for downstream analysis. In the realm of nucleic acid manipulation, sample preparation relates to the steps that occur following initial purification of DNA. Recently, the number of applications that require upstream preparation of nucleic acid samples has grown dramatically. Techniques such as next-generation sequencing, microarrays and library construction all require efficient, uniform and unbiased processing of nuclear material for accurate analysis. As a result, the demand for high quality sample preparation reagents has grown, as well as the need for novel reagents, novel formats of existing reagents and more stringent quality controls.
Next Generation Sequencing
In 2003, the completion of the human genome sequence marked the end of a large international project, but also the beginning of novel sequencing methods. During the following year, the National Human Genome Research Institute (NHGRI) began funding sequencing projects with the goal of lowering the cost of sequencing a human genome from $3 billion (1) down to $100,000 – and ultimately to $1,000. Since then, several strategies for massively parallel sequencing have been commercialized or are in development. These include pyrosequencing (Roche 454), sequencing by synthesis (Illumina, Intelligent Bio-Systems, Helicos BioSciences, LaserGen), sequencing by ligation (Applied Biosystems/Life Technologies, Complete Genomics, Polonator), real-time sequencing (Pacific Biosciences) and nanopore sequencing (Oxford Nanopore Technologies, Sequenom).
In recent years, the advent of these technologies has revolutionized the world of sequencing and biology. There is an insatiable appetite for DNA sequencing across diverse scientific disciplines including microbiology, evolution, ecology, forensics, epidemiology, comparative genomics, and diagnostic applications.
Parallel sequencing methods have enabled whole genome resequencing of human genomes (2,3), analysis of chromatin binding sites along the entire genome by ChIP analysis (4,5), methylation analysis (6), transcriptome analysis (7), metagenomic surveys, and sequencing of ancient DNA (8,9).
Reagents for Sample Preparation
DNA and RNA sample preparation is one of the most important aspects of optimization of the quality and quantity of nucleic acid material for next generation sequencing. The exquisite sensitivity of these technologies places new demands on all the reagents involved, including those required for the upstream sample preparation process, which is outlined in Figure 1. During sample preparation, genomic DNA is sheared into 150 – 800 bp fragments. This can be done by nebulization, acoustic shearing or by treatment with enzymes. DNA sheared by nebulization or acoustic shearing may contain ends that are incompatible (3´ or 5´ overhangs and 3´-phosphates or 5´-OH) with subsequent ligation steps and therefore must be repaired. In some cases, it may be necessary to convert a mixture of heterogeneous ends to a homogenous population of blunt ended DNA fragments with 5´-phosphates and 3´-OH. Furthermore, if dA tailing is required for subsequent ligation, a 3´ dAMP must be added to a blunt ended fragment. Adaptors are then ligated either to blunt ended or dA tailed fragments and the entire library is amplified.
Because sample preparation is a series of coordinated reactions, each step must be as efficient as possible to maximize yield while at the same time maintaining convenience and cost effectiveness. The efficiency of end repair, dA tailing, ligation and amplification are all dependent on the purity of the enzymes that perform these steps.
For 35 years, New England Biolabs has been committed to providing enzymes that drive advances in molecular biology. The breadth of NEB products is built on a foundation of core enzymes that has expanded to include enzymes with unique properties, as well as new applications and combinations of existing enzymes. In fact, scientists have been successfully using NEB reagents for sample preparation of nucleic acid material for many years, and to meet the demands of our customers, NEB offers a broad selection of reagents that facilitate sample preparation of DNA (Figure 2). Additionally, NEB has taken a dual approach to developing products for sample preparation:
- Develop novel solutions for multiple steps in sample preparation processes. For example, the PreCR Repair Mix (NEB #M0309) is an optimized enzyme master mix that repairs damaged DNA and also offers a simplified workflow.
- Offer additional formulations of reagents for increased ease-of-use in specific applications. An example of this approach is the NEBNext® DNA Sample Prep Reagent Set 1, a set of enzymes, nucleotides and buffers ideally suited for sample preparation for next-generation sequencing and expression library construction.
Figure 1: Genomic library construction using NEB reagents†
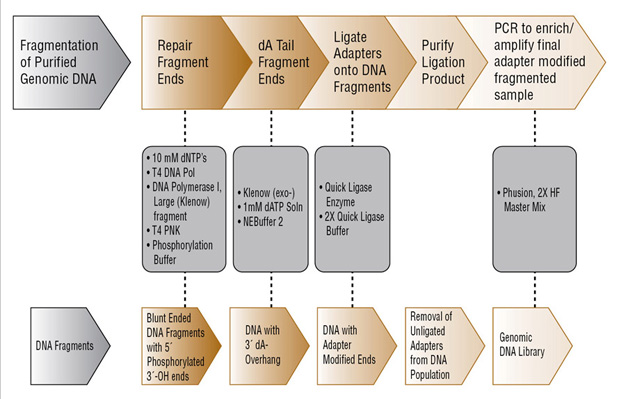
Figure 2: Reagents for DNA Sample Preparation

Quality Assurance
Our priority is to provide product formulations that appropriately address existing and future demanding applications, thereby providing maximum convenience and quality to customers. As new and more sensitive applications develop, quality controls and functional assays must evolve to ensure that the more stringent demands placed on reagents are met. For example, each component of the NEBNext DNA Sample Prep Reagent Set 1 undergoes rigorous quality controls and validation assays. In addition to the standard quality controls for contaminating endonuclease and exonuclease activities, each component also meets further stringent quality controls relevant for these applications, including screening for RNase activity, phosphatase activity, functional activity assays and physical purity. Finally, these reagents are functionally validated together by preparation of a genomic DNA library that is sequenced on an Illumina Genome Analyzer II, and by preparation of an expression library.
Click here for examples of the individual quality control assays performed for Quick T4 DNA Ligase, a component of the NEBNext DNA Sample Prep Reagent Set 1. Specific details of each quality control are supplied on the data card accompanying the product.
For more information regarding the NEBNext product line, please contact NEBNext@neb.com.
Customized Solutions
Additionally, to meet the demands of high-throughput customers, NEB reagents can be supplied in bulk quantities, with customized formulations and supply formats available. NEB is committed to working closely with larger consumption customers at the research, development and production levels to optimize reagent integration specific to technology platforms.
For more information regarding customized solutions and OEM opportunities, please contact custom@neb.com.
New England Biolabs is committed to meeting the current and emerging needs of scientists in this rapidly evolving area of research.
References:
- Collins, F. S., et al. (2003). “The Human Genome Project: Lessons from a large-scale biology.” Science, 300, 286–290.
- Bentley, D. R., et al. (2008). “Accurate whole human genome sequencing using reversible terminator chemistry.” Nature, 456, 53–9.
- Wheeler, D. A., et al. (2008). “The complete genome of an individual by massively parallel DNA sequencing.” Nature, 452, 872–6.
- Johnson, D. S., et al. (2007). “Genome-wide mapping of in vivo protein-DNA interactions.” Science, 316, 1497–502.
- Mikkelsen, T. S., et al. (2007). “Genome-wide maps of chromatin state in pluripotent and lineage-committed cells.” Nature, 448, 553–60.
- Meissner, A., et al. (2008). “Genome-scale DNA methylation maps of pluripotent and differentiated cells.” Nature, 454, 766–70.
- Tang, F., et al. (2009). “mRNA-Seq whole-transcriptome analysis of a single cell.” Nat. Methods, 6, 377–82.
- Ermini, L., et al. (2008). “Complete mitochondrial genome sequence of the Tyrolean Iceman.” Curr. Biol., 18, 1687–93.
- Green, R. E., et al. (2008). “A complete Neandertal mitochondrial genome sequence determined by high-throughput sequencing.” Cell, 134, 416–26.
Scientific Contributors:
Cynthia Henrickson, Ph.D. and Derek Robinson – development of quality controls
From NEB expressions Spring 2009, vol 4.1By Fiona Stewart, Ph.D. and Andrew Gardner, New England Biolabs, Inc.

