Programming Life: Inquiry & Engineering Through Synthetic Biology
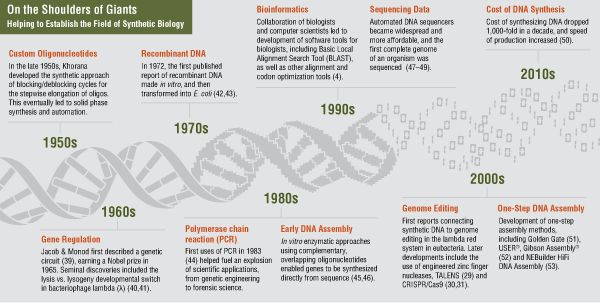
The five decades following the birth of molecular cloning have witnessed an incredible scaling-up of molecular biology due, in large part, to the development of high-throughput technologies in nucleic acid sequencing and macromolecular analysis. But long absent from the resulting explosion of information has been the ability to rationally recreate, in the laboratory, the regulatory complexity of the very gene networks forming the basis cellular behavior. In short, we know a great deal about the “code of life” but are only now beginning to be able to program with it. This aspiration has, in part, given birth to the rapidly developing field of synthetic biology, which aims to unite the rigor of engineering with the design and construction of recombinant nucleic acids, with which to study and understand the behavior of genetic circuits as well as utilize them for technological ends.
Peter Weigle, Ph.D., New England Biolabs, Inc. and Jennifer Redig, Ph.D., BiteSize Bio
What is Synthetic Biology?
Though a comprehensive definition of synthetic biology is elusive, one may characterize it as a “build to understand” approach to biology (3). A quote by the famous theoretical physicist Richard Feynman epitomizes a theme characteristic of the field – “What I cannot create, I do not understand.” How does this sentiment relate to recombinant DNA? Imagine beginning with a repertoire of well-characterized DNA “parts” encoding biological functions such as receptors, promoters, activators, repressors, terminators and reporter genes (or other outputs) – and attempting to rearrange them into configurations designed to direct a biological system (typically, a cellular “chassis”) to accomplish a desired task. Think a pollution detecting E. coli cell that expresses green fluorescent protein (GFP) in the presence of arsenic and then self-destructs after a given period of time, or an engineered implantable human cell line that undergoes a preset number of cell divisions and then secretes insulin at precisely regulated levels in response to extracellular glucose concentrations.
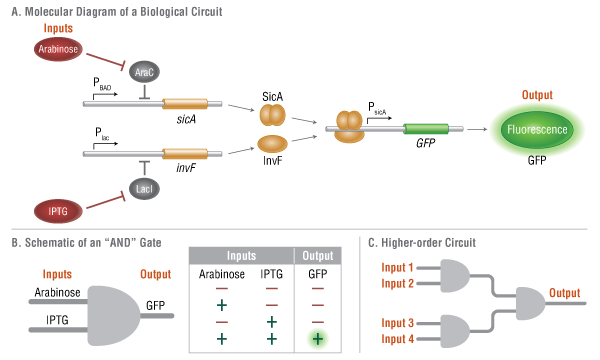
Such re-purposed cells would be described, in synthetic biology parlance, as “genetic devices.” These devices are designed for multi-step behaviors, and relative to earlier examples of genetic engineering, their design is necessarily complex. How cells can be programmed for such functions is neither intuitive nor obvious. Here, synthetic biology has made a radical departure from previous forms of genetic engineering by borrowing engineering concepts from control theory and digital computing as a framework upon which to design genetic circuits for programming cellular behaviors. A genetic implementation of one such “simple” computational operation, the Boolean AND gate, is shown in Figure 1. Higher order combinations of multiple kinds of genetically encoded Boolean operations, and other types of synthetic gene circuits, have been constructed to perform a variety of simple computational tasks, including edge-detection, cell to cell communication, and counting of signal inputs (4).
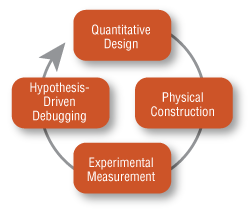
Going from a circuit schematic to a working genetic device is guided by an engineering paradigm: the design-build-test cycle (Figure 2, p. 4). A key tool in this process is computer-aided mathematical modeling. Unlike their electrical counterparts, genetic circuits operate under conditions that dominate the cellular environment. A model attempting to describe and predict the behavior of a genetic device must accurately incorporate a range of parameters such as diffusion, binding equilibria, networks of protein/DNA interactions, and dynamic reactant concentrations; a variety of deterministic and stochastic approaches have been employed to accomplish this goal (5). As such, the model embodies a sophisticated hypothesis about how the device might work. The genetic device is prototyped (e.g., synthetic DNA is assembled and transformed into the cell) and its behavior evaluated in terms of the model. What is learned during each stage is used to improve the performance of the device in subsequent rounds of the cycle – through changes to the device itself, as well as through refinements to the model.
While synthetic biology shares many of the tools and reagents with hypothesis-driven experimental biology and molecular biology, it follows a fundamentally different approach. Many of the techniques in a molecular biologist’s repository (e.g., oligo synthesis, genome editing) may not exist in their current form were it not for synthetic biology. Conversely, synthetic biologists can build upon discoveries made by molecular biologists. In essence, synthetic biologists assemble genetic components in order to execute an “artificial” function, and in the process of getting it to work, the engineered genetic construct becomes itself an object of study and yields basic principles for application to subsequent designs.
Chemical engineering in vivo
A practical definition of synthetic biology must also include the latest developments in industrial fermentation and metabolic engineering. Even a cursory survey of papers and journals covering synthetic biology shows a significant number of reports describing synthetic biology to synthesize fuels, chemicals and materials. Historically, this technology began as an outgrowth of beer and wine making, after it was discovered that fermentation could also be used to produce economically valuable solvents and organic acids (6). With the advent of greatly expanded sequence databases, inexpensive DNA synthesis, and genome engineering methods, it has become increasingly practical to do chemical synthesis in vivo. Designer metabolic pathways utilizing genes encoding enzymes derived from any of the domains of life can inserted into microbes such as Saccharomyces cerevisiae or E. coli, endowing them with the ability to convert cheap chemical inputs, such as starch- or cellulose-derived sugars, into more commercially valuable chemicals.
The First Synthetic Gene Circuits
In 2000, the first synthetic circuits were made
when Gardner, Cantor and Collins created a genetic
toggle switch (12), and Elowitz and Leibler
engineered a repressilator, a synthetic genetic regulatory
network designed from scratch to produce
stable oscillations of gene expression (13). Both
of these circuits were model-based, but both also
needed experimental fine-tuning to achieve agreement
between model and experimental output.
These experiments were quickly followed by “The
First International Meeting on Synthetic Biology”
or SB1.0, which was held in 2004 at MIT
(14). Attended by biologists, chemists, physicists,
engineers and computer scientists, the goal of this
conference was to bring together those scientists
interested in creating and characterizing synthetic
biological systems. This meeting, and smaller ones
like it, laid down the foundation of a new, emerging
discipline.
A Community to Build From
As the synthetic biology discipline grew, it quickly became clear that there needed to be a more efficient way to assemble genetic parts and circuits (4). Without established methods for assembly and testing, researchers were forced to ad hoc experimental designs, wasting time and money by designing, testing and redesigning constructs. To combat these issues, a public repository, the Registry of Standard Biological Parts (RSBP), was founded at MIT by Tom Knight and Drew Endy. The goal of this repository is to catalog and develop genetic parts into ‘BioBricks®’ that could be used for the assembly of larger circuits. The Bio- Brick standard was developed to ensure that parts could be easily shared and used among synthetic biologists by requiring submitted parts to conform to a simplified cloning scheme utilizing four restriction enzymes. However, it became quickly clear that the task of populating the Registry with biological parts, and the work of characterizing them to establish their utility, would dwarf the resources of the relatively small numbers of labs devoted to synthetic biology. Out of this daunting mission, and the need to sustainably train a new generation of synthetic biologists, the International Genetically Engineered Machine (iGEM) competition was born (15).
Training the next generation of bioengineers
Since its inception in 2004, iGEM has evolved into a highly successful vehicle for training and showcasing a new generation of biological engineers using the synthetic biology framework. In 2014, iGEM hosted its 10th annual Jamboree, with over 4,000 participants from across the globe presenting projects that detailed their efforts to model, build and test genetic devices. Students competed in a variety of tracks such as Food, Medicine, Manufacturing and Information Processing. Each team was also asked to demonstrate that they have considered the impact and implications of the technologies they are developing through dialog with relevant stakeholders. Teams were supported by various organizations, including NEB. To date, more than 28,000 student competitors have participated in this engineering competition.
A rapidly maturing field
Synthetic Biology as a discipline continues to
grow rapidly. Recent synthetic biology developments
include:
Circuits Get Complex – In the early 2000s, DNA
circuits continued to advance. More elements were
added (16), and sensing became more diversified
(17,18). Additionally, RNA, not just DNA, was
used in circuit generation (19–21).
‘Synthetic’ Used to Investigate ‘Native’ – Beginning
in 2009, designed circuits were used to understand
native systems through compare/contrast
schemes (22) of engineered versus native systems.
Synthetic Biology was not just limited to engineering
new biology; it was also used to investigate and
understand native biology.
Commercially Valuable Products are Made –
In the 2000s, amino acid biosynthesis was used to
produce commercially valuable products such as
isobutanol (23,24), biodiesel (25) and gasoline (26).
These experiments were a logical extension of fermentation
biotechnology and highlighted synthetic
biology’s commercial and environmental promise.
Assembly of a Whole Bacterial Genome in Yeast
– In 2008, researchers were able to take advantage
of yeast’s remarkable ability to recombine overlapping
DNA fragments to assemble an entire genome
in a single step. This method allowed for speedier
assembly of DNA molecules than previous methods
(27,28).
New Genome Editing Tools Emerge – Beginning
in 2010, zinc finger nucleases gave way to more
precise genome editing tools, from TALENS (29) to,
in 2013, CRISPR/Cas9 (30) systems. This empowered
synthetic and molecular biologists to create and
explore as never before. In addition, a catalytically
inactive form of Cas9, known as dCas9, has further
enhanced the usefulness of the CRISPR/Cas9 system
by enabling both activation and repression of
transcription in yeast and mammalian cells, allowing
modulation of endogenous gene expression (31).
First “Artificial Cell” Engineered – In 2010, Craig
Venter and colleagues demonstrated just how far
the discipline of synthetic biology had come when
they published a paper disclosing the recreation of a
Mycoplasma mycoides cell controlled by a chemically-synthesized
genome (32).
Therapies Engineered – In 2010, Fussenegger
and colleagues engineered a synthetic circuit that,
when inserted into the genome of a mouse mutant
bred to develop hyperuricemia, was able to maintain
uric acid homeostasis, essentially correcting an
inborn metabolic defect (33). This demonstrated
the therapeutic promise of synthetic biology.
Ongoing Challenges
Synthetic Biology is a young field, but it has
achieved much in a short time period. However,
like all disciplines, it continues to face challenges.
Measurement, Robustness and Predictability
– Aspects of synthetic biology still remain an art.
Genetic circuits often require much “tweaking”
in order to get them to function in the context
for which they were designed. Further principles
governing the function of genetic circuits will have
to be elucidated to improve the interoperability of
genetic parts in multiple contexts.
Cells are Not Exactly Digital – Though incredibly
powerful as a guiding framework for designing,
building, and testing genetic circuits, the digital
circuit metaphor has limits. Biological systems
differ from electronic ones in fundamental ways,
and modeling genetic regulation remains under
determined. Synthetic biology researchers continue
to incorporate new ideas and theories to describe,
model and predict genetic circuit behaviors. A new
and promising area utilizes analogies to analog
circuitry (34).
Ethics and Safety – Synthetic Biology, and indeed
all genetic engineering, has provoked concern over
potential misuse, intentional or accidental. There
is active discussion regarding potential impacts
(35). Built-in forms of biological containment are
also an active area of investigation, including the
refinement of genetic “kill switches”, which ideally
would ensure that genetic devices could not
survive outside of the laboratory or factory. Government
policy has and will continue to weigh in:
information on the ethics of synthetic biology can
be found in the 2010 Presidential Bioethics Commission
report on synthetic biology (36). As with
other technologies, a scientifically literate public is
a requirement for nuanced and effective dialog.
Future Directions
The past sixty years have seen incredible scientific and technological advances based on the ability to compose in DNA. DNA-driven technologies will continue to absorb developments and ways of thinking from diverse fields. Advances in materials sciences, nanotechnology, microfluidics, automated liquid handling, indeed all the applied sciences, will drive new applications using cellular systems and even biological technologies beyond the cell (37,38). The proliferation and use of these technologies will continue to impact our lives. With prudence and foresight, they may prove indispensible to our survival.