NEBNext Library Quant Kit Protocol - NEBNext Library Quant Kit (E7630)
Symbols
![]() This caution sign signifies a step in the protocol that has multiple paths leading to the same end point but is dependent on a user variable, like the amount of input DNA.
This caution sign signifies a step in the protocol that has multiple paths leading to the same end point but is dependent on a user variable, like the amount of input DNA.
Starting Material: 5 ng–1 μg fragmented DNA
3.1. Thaw and Mix Reagents
3.1.1. Thaw the NEBNext Library Quant Master Mix and NEBNext Library Quant Primer Mix. Ensure mixing of NEBNext Library Quant Primer Mix by vortexing for 5 seconds. Centrifuge briefly to collect material from the sides of the tubes. Place reagents on ice.
3.1.2. Thaw the NEBNext Library Quant DNA Standards, tubes 1–4. Mix well by inverting 3–5 times. Centrifuge briefly to collect material from the sides of the tubes. Place on ice.
3.1.3. Thaw the NEBNext Library Quant Dilution Buffer (10X). Mix well by vortexing 10 seconds. Centrifuge briefly to collect material from the sides of the tube. Place on ice.
3.2. Preparation of NEBNext Library Quant Master Mix (with primers)
Prepare the NEBNext Library Quant Master Mix (with primers) by adding 100 μl NEBNext Library Quant Primer Mix to a tube of NEBNext Library Quant Master Mix (1.5 ml; NEB #E7630S ) or 500 μl NEBNext Library Quant Primer Mix to a bottle of NEBNext Library Quant Master Mix (7.5 ml; NEB #E7630L ). Mix by vortexing for 10 seconds.
Record the date on the NEBNext Library Quant Master Mix container to indicate that the NEBNext Library Quant Primer Mix has been added.
This mix can be stored at 4°C for convenience for up to six weeks. For longer term storage, the mix will be stable for seven months at –20°C and for 30 freeze/thaw cycles.
ROX: If using a real-time instrument that uses ROX normalization dye, we
recommend adding ROX to the NEBNext Library Quant Master Mix after
thawing. Both a low and high concentration of ROX are provided at 100X
concentration (refer to Table 1 for ROX recommendations for common qPCR
instruments).
For E7630S add 20 μl of ROX to the NEBNext Library Quant Master Mix tube, and for E7630L add 100 μl.
Alternatively, ROX can be added to each reaction if desired (0.2 µl per well in 96 well format and 0.1 µl per well in 384 well format).
Dilute the NEBNext Library Quant Dilution Buffer (10X) 1:10 with nuclease-free water. Mix by vortexing for 10 seconds. Prepare sufficient buffer for the desired number of libraries to be quantitated, allowing 1.2 ml for each library.
| NEBNext Library Quant Dilution Buffer | 1 ml |
| Nuclease-free Water | 9 ml |
| Total volume | 10 ml |
3.4. Dilution of Library Samples
Perform an initial 1:1,000 dilution of each library sample in 1X NEBNext Library Quant Dilution Buffer in a nuclease-free tube as described below. Pipet carefully and mix thoroughly by vortexing for 10 seconds. Centrifuge briefly to collect material from the sides of the tube.
3.4.1. Add 1 μl library to 999 μl NEBNext Library Quant Dilution Buffer to create a 1:1,000 dilution.
Prepare two additional dilutions of the library sample to create 1:10,000 and 1:100,000 dilutions as described below. Mix each thoroughly by vortexing for 10 seconds. Centrifuge briefly to collect material from the sides of the tube. These library dilutions will be used for qPCR analysis.
3.4.2. Add 10 μl of the 1:1,000 dilution from Step 1 to 90 μl of 1X NEBNext Library Quant Dilution Buffer to create a 1:10,000 dilution.
3.4.3. Add 10 μl of the 1:10,000 dilution from Step 2 to 90 μl of 1X NEBNext Library Quant Dilution Buffer to create a 1:100,000 dilution.
The range of standard concentrations in the NEBNext Library Quant Kit is 10–0.01 pM. Most library prep protocols will produce libraries with concentrations of 1–200 nM, thus the 1:100,000 dilution will fall in range of the standards for nearly all libraries (e.g. 2 pM, from a 1:100,000 dilution of 200 nM, will amplify near standard 2 in the qPCR run). Libraries prepared with very low yield will still be quantitated by the kit, with the 1:10,000 and 1:100,000 dilution recommended for libraries expected to be as low as 0.2 nM. The 1:1,000 dilution can be run for lower concentration libraries.
For best results, we recommend running each DNA standard and library sample in triplicate.
3.5.1. Prepare DNA standards and diluted library samples.
| NEBNext Library Quant Master Mix (with primers) | 16 μl |
| DNA standard or library dilution | 4 μl |
| Total volume | 20 μl |
3.5.2. We recommend inclusion of a no template control (NTC) reaction in addition to the 4 DNA Standards. The Cq from the NTC will not be used in quantitation analysis, but serves as a valuable control reaction to ensure performance of the kit and absence of sample contamination. Prepare a no-template control as follows:
| NEBNext Library Quant Master Mix (with primers) | 16 μl |
| Library Dilution Buffer (1X) | 4 μl |
| Total volume | 20 μl |
Mix reactions by pipetting sample or buffer at least 5X. Try to minimize bubbles in plate wells, but 1–2 bubbles per well on the top of the liquid will be removed by heating and not affect results.
If using a 384-well or other instrument with small reaction volumes (e.g. 10 μl) then simply reduce the above volumes accordingly. For 10 μl reactions use 8 μl of NEBNext Library Quant Master Mix (with primers) and 2 μl of NEBNext Library Quant DNA Standards, library dilution or NEBNext Library Quant Dilution Buffer:
| NEBNext Library Quant Master Mix (with primers) | 8 μl |
| DNA standard, library dilution or dilution buffer | 2 μl |
| Total volume | 10 μl |
3.6. Load the qPCR Plate
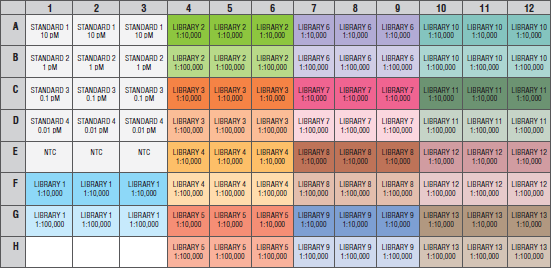
Prepare the qPCR plate as desired, with a recommended layout shown below.
- If reactions were not prepared in the final qPCR plate, transfer them into the appropriate plate for the real-time instrument being used.
- Although most bubbles will pop during thermal cycling, it is best to avoid bubble formation.
- If desired, include a no template control (NTC) assay in the plate in triplicate.
- Include qPCR assays for the 4 prediluted NEBNext Library Quant DNA Standards 1–4 in triplicate.
- Include qPCR assays for at least 2 dilutions of each library sample (1:10,000 and 1:100,000), with 3 dilutions each if desired.
Note: Standards and NTC require 15 wells of the plate, leaving 81 for libraries. This enables quantitation of 9 libraries with 3 dilutions each loaded on the plate, or 13 libraries with 2 dilutions. - Seal the qPCR plate, and if desired, centrifuge at 2500–3000 rpm for 2 minutes to collect material at the bottom of wells.
3.7. Run the qPCR Plate in a Real-time Thermal Cycler
3.7.1. Run the qPCR assay following the cycling conditions listed below:
|
CYCLE STEP |
TEMP | TIME | CYCLES |
| Initial Denaturation | 95°C | 1 Minute | 1 |
| Denaturation Extension |
95°C 63°C |
15 seconds 45 seconds |
35 |
Ensure a plate read is included at the end of the extension step. Use the SYBR or SYBR/FAM setting on the real-time instrument. When ROX is used as normalization dye, verify that ROX is selected in the appropriate setting.
We recommend the “Fast” temperature cycling profile where applicable (e.g. Applied Biosystems StepOnePlus, 7500 Fast). The kit can also be run in “Standard” temperature mode if desired, but this will result in a longer time required for the PCR protocol to be completed.

