Reaction Conditions for Chemical Coupling (S9153)
Example Reaction: Coupling of BG-Maleimide to a 5´ thiol modified Oligonucleotide:
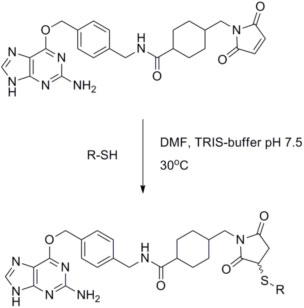
Figure 1. Reaction of BG-Maleimide with thiol groups. The 5´-thiol modified oligonucleotide (43 nmol) was reduced by incubation for 1 hour at room temperature with 10 mM DTT in 200 µl 20 mM Tris-HCl pH 8.5. The DTT was removed by gel filtration and the oligonucleotide eluted in PBS buffer (pH 7.4). The most concentrated fractions were combined giving a total of 800 µl. Three hundred microliters of BG-maleimide solution (2.5 mM in DMF) was added and the reaction mixture incubated at room temperature for 1 hour. The reaction mixture was diluted with water to a total volume of 2 ml and excess maleimide removed by gel filtration. The BG-maleimide-oligonucleotide conjugate was then purified by HPLC (solvent A: 0.1 M tetraethylammonium acetate pH 6.9 in water; solvent B: acetonitrile).

